IRGQ-mediated autophagy in MHC class I quality control promotes tumor immune evasion
IF 45.5
1区 生物学
Q1 BIOCHEMISTRY & MOLECULAR BIOLOGY
引用次数: 0
Abstract
The autophagy-lysosome system directs the degradation of a wide variety of cargo and is also involved in tumor progression. Here, we show that the immunity-related GTPase family Q protein (IRGQ), an uncharacterized protein to date, acts in the quality control of major histocompatibility complex class I (MHC class I) molecules. IRGQ directs misfolded MHC class I toward lysosomal degradation through its binding mode to GABARAPL2 and LC3B. In the absence of IRGQ, free MHC class I heavy chains do not only accumulate in the cell but are also transported to the cell surface, thereby promoting an immune response. Mice and human patients suffering from hepatocellular carcinoma show improved survival rates with reduced IRGQ levels due to increased reactivity of CD8+ T cells toward IRGQ knockout tumor cells. Thus, we reveal IRGQ as a regulator of MHC class I quality control, mediating tumor immune evasion.
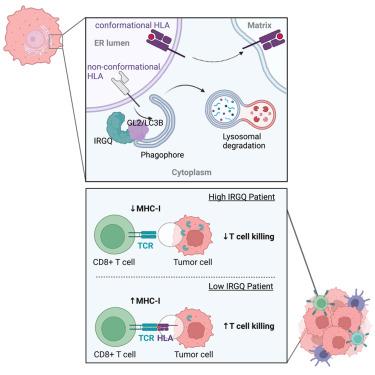
MHC I 类质量控制中 IRGQ 介导的自噬促进肿瘤免疫逃避
自噬-溶酶体系统引导着多种货物的降解,也参与了肿瘤的进展。在这里,我们发现免疫相关 GTPase 家族 Q 蛋白(IRGQ)--一种迄今为止尚未定性的蛋白质--在主要组织相容性复合体 I 类(MHC I 类)分子的质量控制中发挥作用。IRGQ 通过其与 GABARAPL2 和 LC3B 的结合模式将折叠错误的 MHC I 类导向溶酶体降解。在缺乏 IRGQ 的情况下,游离的 MHC I 类重链不仅会在细胞内积聚,还会被运输到细胞表面,从而促进免疫反应。由于 CD8+ T 细胞对 IRGQ 基因敲除肿瘤细胞的反应性增加,小鼠和人类肝细胞癌患者在 IRGQ 水平降低的情况下生存率有所提高。因此,我们揭示了 IRGQ 是 MHC I 类质量控制的调节因子,可介导肿瘤免疫逃避。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊
Cell
生物-生化与分子生物学
CiteScore
110.00
自引率
0.80%
发文量
396
审稿时长
2 months
期刊介绍:
Cells is an international, peer-reviewed, open access journal that focuses on cell biology, molecular biology, and biophysics. It is affiliated with several societies, including the Spanish Society for Biochemistry and Molecular Biology (SEBBM), Nordic Autophagy Society (NAS), Spanish Society of Hematology and Hemotherapy (SEHH), and Society for Regenerative Medicine (Russian Federation) (RPO).
The journal publishes research findings of significant importance in various areas of experimental biology, such as cell biology, molecular biology, neuroscience, immunology, virology, microbiology, cancer, human genetics, systems biology, signaling, and disease mechanisms and therapeutics. The primary criterion for considering papers is whether the results contribute to significant conceptual advances or raise thought-provoking questions and hypotheses related to interesting and important biological inquiries.
In addition to primary research articles presented in four formats, Cells also features review and opinion articles in its "leading edge" section, discussing recent research advancements and topics of interest to its wide readership.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


