An entry to spiro-fused sultams via electrochemical brominative ipso-annulation of N-aryl alkynyl sulphonamides
IF 16.4
1区 化学
Q1 CHEMISTRY, MULTIDISCIPLINARY
引用次数: 0
Abstract
Synthesis of cyclohexadienone spiro-fused sultams is established through electrochemical brominative intramolecular ipso-annulation of N-aryl alkynyl sulphonamides. A range of aryl/heteroarylated-alkynyl sulphonamides are suitable under external oxidant-free electrochemical conditions. Lithium bromide is used as bromine source in this domino bromination/ipso-cyclization, leading to the corresponding bromo spiro-sultams in good yields.
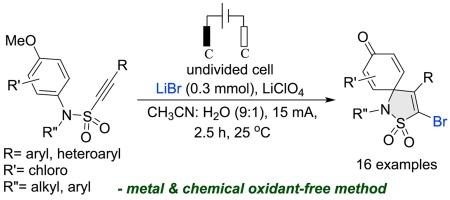
通过 N-芳基炔基磺酰胺的电化学溴化异烷基化反应进入螺融舒坦类化合物领域
通过对 N-芳基炔基磺酰胺进行电化学溴化分子内异annulation,合成了环己二烯酮螺融舒坦。在外部无氧化剂的电化学条件下,一系列芳基/异芳基炔基磺酰胺均适用。在这种多米诺溴化/异环化反应中,溴化锂被用作溴源,从而以良好的收率得到相应的溴代螺-苏丹。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

Accounts of Chemical Research
化学-化学综合
CiteScore
31.40
自引率
1.10%
发文量
312
审稿时长
2 months
期刊介绍:
Accounts of Chemical Research presents short, concise and critical articles offering easy-to-read overviews of basic research and applications in all areas of chemistry and biochemistry. These short reviews focus on research from the author’s own laboratory and are designed to teach the reader about a research project. In addition, Accounts of Chemical Research publishes commentaries that give an informed opinion on a current research problem. Special Issues online are devoted to a single topic of unusual activity and significance.
Accounts of Chemical Research replaces the traditional article abstract with an article "Conspectus." These entries synopsize the research affording the reader a closer look at the content and significance of an article. Through this provision of a more detailed description of the article contents, the Conspectus enhances the article's discoverability by search engines and the exposure for the research.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


