Relationship between inflammation/immunity and epilepsy: A multi-omics mendelian randomization study integrating GWAS, eQTL, and mQTL data
IF 2.3
3区 医学
Q2 BEHAVIORAL SCIENCES
引用次数: 0
Abstract
Objectives
Increasing evidence suggests that activated innate/adaptive immunity induces an inflammatory response, thereby participating in epileptogenesis. However, the biological explanation of inflammation/immunity as a potential cause for epilepsy remains largely unknown. This research aimed to determine the causal effects of inflammation/immune-related genes in epilepsy based on multi-omics mendelian randomization (MR).
Methods
We employed summary-data-based MR (SMR) approach to combine GWAS for epilepsy (12,891 cases and 312,803 control) with gene expression quantitative trait loci (cis-eQTL, 31,684 participants) and DNA methylation QTL (cis-mQTL, 1,980 participants) data. Five additional MR methods were then used for sensitivity analyses to confirm the reliability of causal associations. In addition, enrichment analysis of key genes was conducted to provide insight into the biological functions of epilepsy risk variants.
Results
A total of 386 inflammation/immune-related genes were selected for further analyses. Primary SMR analysis indicated that 37 DNA methylation sites and six genes regulated by them had potential causal relationship with epilepsy. MR analysis further refined the results, identifying three genes that had a causal effect on epilepsy. Notably, VEGFA (OR: 0.925; 95 % CI: 0.862–0.994) expression was negatively correlated with epilepsy risk, whereas IL16 (OR: 1.076; 95 % CI: 1.028–1.126) and HLA-DPA1 (OR: 1.041; 95 % CI: 1.009–1.074) expressions were positively associated with epilepsy risk. Functional enrichment analysis revealed that the identified genes were involved in GO-BP terms related to VEGF activation signaling and chemotaxis regulation.
Conclusion
This analysis confirms the causal role of inflammation/immunity in epilepsy, and the identified candidate genes provide clues for drug development in clinical practice.
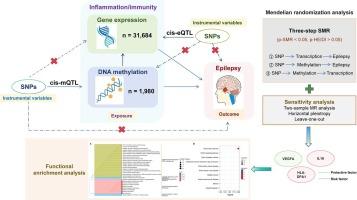
炎症/免疫与癫痫之间的关系:整合全球基因组研究、eQTL 和 mQTL 数据的多组学随机研究
目的:越来越多的证据表明,激活的先天/适应性免疫会诱发炎症反应,从而参与癫痫的发生。然而,炎症/免疫作为癫痫潜在病因的生物学解释在很大程度上仍然未知。本研究旨在基于多组学门德尔随机化(MR)确定炎症/免疫相关基因在癫痫中的因果效应:我们采用了基于汇总数据的MR(SMR)方法,将癫痫的GWAS(12,891例病例和312,803例对照)与基因表达定量性状位点(顺式-eQTL,31,684名参与者)和DNA甲基化QTL(顺式-mQTL,1,980名参与者)数据相结合。然后使用另外五种磁共振方法进行敏感性分析,以确认因果关联的可靠性。此外,还对关键基因进行了富集分析,以深入了解癫痫风险变异的生物学功能:结果:共选取了386个炎症/免疫相关基因进行进一步分析。初级 SMR 分析表明,37 个 DNA 甲基化位点及其调控的 6 个基因与癫痫有潜在的因果关系。磁共振分析进一步完善了结果,确定了三个与癫痫有因果关系的基因。值得注意的是,VEGFA(OR:0.925;95 % CI:0.862-0.994)的表达与癫痫风险呈负相关,而IL16(OR:1.076;95 % CI:1.028-1.126)和HLA-DPA1(OR:1.041;95 % CI:1.009-1.074)的表达与癫痫风险呈正相关。功能富集分析表明,鉴定出的基因参与了与血管内皮生长因子激活信号转导和趋化调节相关的GO-BP术语:结论:这项分析证实了炎症/免疫在癫痫中的因果作用,所发现的候选基因为临床实践中的药物开发提供了线索。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

Epilepsy & Behavior
医学-行为科学
CiteScore
5.40
自引率
15.40%
发文量
385
审稿时长
43 days
期刊介绍:
Epilepsy & Behavior is the fastest-growing international journal uniquely devoted to the rapid dissemination of the most current information available on the behavioral aspects of seizures and epilepsy.
Epilepsy & Behavior presents original peer-reviewed articles based on laboratory and clinical research. Topics are drawn from a variety of fields, including clinical neurology, neurosurgery, neuropsychiatry, neuropsychology, neurophysiology, neuropharmacology, and neuroimaging.
From September 2012 Epilepsy & Behavior stopped accepting Case Reports for publication in the journal. From this date authors who submit to Epilepsy & Behavior will be offered a transfer or asked to resubmit their Case Reports to its new sister journal, Epilepsy & Behavior Case Reports.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


