Multiparameter imaging reveals clinically relevant cancer cell-stroma interaction dynamics in head and neck cancer
IF 45.5
1区 生物学
Q1 BIOCHEMISTRY & MOLECULAR BIOLOGY
引用次数: 0
Abstract
Epithelial tumors are characterized by abundant inter- and intra-tumor heterogeneity, which complicates diagnostics and treatment. The contribution of cancer-stroma interactions to this heterogeneity is poorly understood. Here, we report a paradigm to quantify phenotypic diversity in head and neck squamous cell carcinoma (HNSCC) with single-cell resolution. By combining cell-state markers with morphological features, we identify phenotypic signatures that correlate with clinical features, including metastasis and recurrence. Integration of tumor and stromal signatures reveals that partial epithelial-mesenchymal transition (pEMT) renders disease outcome highly sensitive to stromal composition, generating a strong prognostic and predictive signature. Spatial transcriptomics and subsequent analyses of cancer spheroid dynamics identify the cancer-associated fibroblast-pEMT axis as a nexus for intercompartmental signaling that reprograms pEMT cells into an invasive phenotype. Taken together, we establish a paradigm to identify clinically relevant tumor phenotypes and discover a cell-state-dependent interplay between stromal and epithelial compartments that drives cancer aggression.
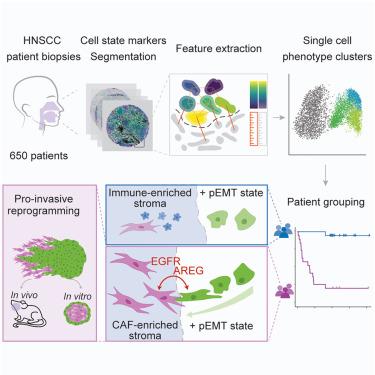
多参数成像揭示头颈癌中与临床相关的癌细胞-基质相互作用动态
上皮肿瘤的特点是肿瘤间和肿瘤内存在大量异质性,这使得诊断和治疗变得复杂。人们对肿瘤与基质之间的相互作用对这种异质性的影响知之甚少。在这里,我们报告了一种以单细胞分辨率量化头颈部鳞状细胞癌(HNSCC)表型多样性的范例。通过将细胞状态标记物与形态特征相结合,我们确定了与临床特征(包括转移和复发)相关的表型特征。整合肿瘤和基质特征后发现,部分上皮-间质转化(pEMT)使疾病结果对基质组成高度敏感,从而产生了一个强大的预后和预测特征。空间转录组学和随后的癌症球体动力学分析确定了癌症相关成纤维细胞-pEMT 轴是室间信号转导的枢纽,它将 pEMT 细胞重编程为侵袭性表型。总之,我们建立了一个范例来识别临床相关的肿瘤表型,并发现了基质和上皮细胞间依赖细胞状态的相互作用,这种相互作用推动了癌症的侵袭。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊
Cell
生物-生化与分子生物学
CiteScore
110.00
自引率
0.80%
发文量
396
审稿时长
2 months
期刊介绍:
Cells is an international, peer-reviewed, open access journal that focuses on cell biology, molecular biology, and biophysics. It is affiliated with several societies, including the Spanish Society for Biochemistry and Molecular Biology (SEBBM), Nordic Autophagy Society (NAS), Spanish Society of Hematology and Hemotherapy (SEHH), and Society for Regenerative Medicine (Russian Federation) (RPO).
The journal publishes research findings of significant importance in various areas of experimental biology, such as cell biology, molecular biology, neuroscience, immunology, virology, microbiology, cancer, human genetics, systems biology, signaling, and disease mechanisms and therapeutics. The primary criterion for considering papers is whether the results contribute to significant conceptual advances or raise thought-provoking questions and hypotheses related to interesting and important biological inquiries.
In addition to primary research articles presented in four formats, Cells also features review and opinion articles in its "leading edge" section, discussing recent research advancements and topics of interest to its wide readership.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


