Antimony(V) sorption and coprecipitation with ferrihydrite: An examination of retention mechanisms and the selectivity of commonly-applied extraction procedures
IF 12.2
1区 环境科学与生态学
Q1 ENGINEERING, ENVIRONMENTAL
引用次数: 0
Abstract
We investigated the mechanisms that control Sb(V) sorption and coprecipitation with ferrihydrite across a range of Sb(V) loadings, and examined the associated effects on Sb(V) extractability during the commonly-applied 1 M HCl extraction scheme and the BCR and Wenzel sequential extraction schemes. EXAFS spectroscopy reveals that Sb(V) sorption and coprecipitation mainly involved Sb(V) incorporation into the ferrihydrite structure via edge sharing and double-corner sharing between SbO6 and FeO6 octahedra. Large amounts of these linkages partially stabilized ferrihydrite against extraction with 1 M HCl. Negligible (<0.5%) ferrihydrite-bound Sb(V) was recovered in the “acid extractable” and “reducible” fractions of the BCR scheme, while 1-16% was recovered in the “oxidizable” fraction. As such, the BCR scheme risks ferrihydrite-bound Sb(V) being misidentified as Sb residing mainly in “residual” phases. In contrast, in the Wenzel scheme, almost all sorbed- and coprecipitated-Sb(V) was recovered in the “amorphous hydrous oxide-bound” fraction, with only 0.6-3.3% in the “specifically-bound” fraction (consistent with our finding of Sb(V) retention via incorporation into ferrihydrite, as opposed to adsorption by the ferrihydrite surface). Collectively, the results provide new insights into the retention mechanisms and extraction behaviour of ferrihydrite-bound Sb(V), enhancing our ability to assess Sb contamination in soils, sediments and geogenic wastes.
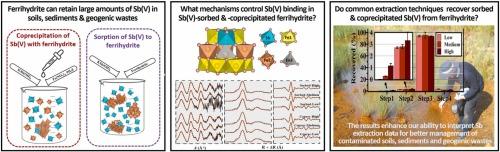
锑(V)与铁酸盐的吸附和共沉淀:对常用萃取程序的保留机制和选择性的研究
我们研究了在不同的 Sb(V)负载范围内控制 Sb(V)吸附和与铁水盐共沉淀的机制,并考察了在常用的 1 M HCl 萃取方案以及 BCR 和 Wenzel 顺序萃取方案中对 Sb(V)萃取率的相关影响。EXAFS 光谱显示,Sb(V) 的吸附和共沉淀主要是通过 SbO6 和 FeO6 八面体之间的边缘共享和双角共享将 Sb(V) 加入到铁水物结构中。这些链接的大量存在部分稳定了铁水物,使其免受 1 M HCl 的萃取。在 BCR 方案的 "可酸萃取 "和 "可还原 "馏分中回收了微不足道(0.5%)的铁镁结合的 Sb(V),而在 "可氧化 "馏分中回收了 1-16%。因此,BCR 方案有可能将铁水结合的 Sb(V)误认为主要存在于 "残余 "相中的 Sb。与此相反,在文泽尔方案中,几乎所有吸附和共沉淀的 Sb(V)都在 "无定形水氧化物结合 "部分中被回收,只有 0.6-3.3% 在 "特异性结合 "部分中(与我们的发现一致,即 Sb(V)是通过融入铁水物而不是被铁水物表面吸附来保留的)。总之,这些结果为我们了解铁水物结合的锑(V)的保留机制和萃取行为提供了新的视角,提高了我们评估土壤、沉积物和地质废弃物中锑污染的能力。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

Journal of Hazardous Materials
工程技术-工程:环境
CiteScore
25.40
自引率
5.90%
发文量
3059
审稿时长
58 days
期刊介绍:
The Journal of Hazardous Materials serves as a global platform for promoting cutting-edge research in the field of Environmental Science and Engineering. Our publication features a wide range of articles, including full-length research papers, review articles, and perspectives, with the aim of enhancing our understanding of the dangers and risks associated with various materials concerning public health and the environment. It is important to note that the term "environmental contaminants" refers specifically to substances that pose hazardous effects through contamination, while excluding those that do not have such impacts on the environment or human health. Moreover, we emphasize the distinction between wastes and hazardous materials in order to provide further clarity on the scope of the journal. We have a keen interest in exploring specific compounds and microbial agents that have adverse effects on the environment.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


