Fast Lithium Ion Conduction in Arsenides Li9AlAs4 and Li9GaAs4
IF 7
2区 材料科学
Q2 CHEMISTRY, PHYSICAL
引用次数: 0
Abstract
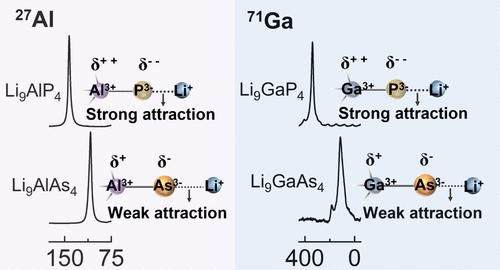
Understanding the structure–property relationship in ionic conductors is crucial for developing more efficient solid-state electrolytes and improving cell architecture. Here, we present two new arsenide-based fast ion conductors, Li9AlAs4 and Li9GaAs4, and compare them with two previously reported phosphide-based superionic conductors, Li9AlP4 and Li9GaP4, to study the substitution effect of element P by As. It is established that anion substitution dramatically enhances lithium-ion mobility and conductivity. In particular, the ionic conductivity and diffusion coefficient at room temperature of Li9GaAs4 reached remarkable levels of 6.5 mS cm–1 and 2.05 × 10–11 m2 s–1, respectively, achieving an order of magnitude increase compared with Li9GaP4. Multinuclear solid-state NMR chemical shifts reveal that As3– in [TrAs4]9– (Tr = Al, Ga) has a lower negative charge density than P3– in [TrP4]9–, which leads to a smaller Coulomb force between Li+ and As3– than between Li+ and P3–. This weakened Coulomb force on lithium ions, caused by As substitution, together with an enlarged lattice volume, lowers the activation barrier and promotes Li ion conductivity.
砷化物 Li9AlAs4 和 Li9GaAs4 中的快速锂离子传导
了解离子导体的结构-性能关系对于开发更高效的固态电解质和改进电池结构至关重要。在此,我们介绍了两种新型砷化物基快速离子导体 Li9AlAs4 和 Li9GaAs4,并将它们与之前报道的两种磷化物基超离子导体 Li9AlP4 和 Li9GaP4 进行了比较,以研究 As 对 P 元素的替代效应。结果表明,阴离子取代能显著提高锂离子的迁移率和电导率。其中,Li9GaAs4 在室温下的离子电导率和扩散系数分别达到了 6.5 mS cm-1 和 2.05 × 10-11 m2 s-1 的显著水平,比 Li9GaP4 提高了一个数量级。多核固态核磁共振化学位移显示,[TrAs4]9-(Tr = Al、Ga)中的 As3- 的负电荷密度低于[TrP4]9-中的 P3-,这导致 Li+ 与 As3- 之间的库仑力小于 Li+ 与 P3- 之间的库仑力。As 取代导致锂离子受到的库仑力减弱,再加上晶格体积增大,从而降低了活化势垒,提高了锂离子的导电性。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

Chemistry of Materials
工程技术-材料科学:综合
CiteScore
14.10
自引率
5.80%
发文量
929
审稿时长
1.5 months
期刊介绍:
The journal Chemistry of Materials focuses on publishing original research at the intersection of materials science and chemistry. The studies published in the journal involve chemistry as a prominent component and explore topics such as the design, synthesis, characterization, processing, understanding, and application of functional or potentially functional materials. The journal covers various areas of interest, including inorganic and organic solid-state chemistry, nanomaterials, biomaterials, thin films and polymers, and composite/hybrid materials. The journal particularly seeks papers that highlight the creation or development of innovative materials with novel optical, electrical, magnetic, catalytic, or mechanical properties. It is essential that manuscripts on these topics have a primary focus on the chemistry of materials and represent a significant advancement compared to prior research. Before external reviews are sought, submitted manuscripts undergo a review process by a minimum of two editors to ensure their appropriateness for the journal and the presence of sufficient evidence of a significant advance that will be of broad interest to the materials chemistry community.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


