Structurally Minimalized and Druglike TGase2 Inhibitors Based on 7-Aminoquinoline-5,8-dione Scaffolds for the Treatment of Diabetic Retinopathy
IF 6.8
1区 医学
Q1 CHEMISTRY, MEDICINAL
引用次数: 0
Abstract
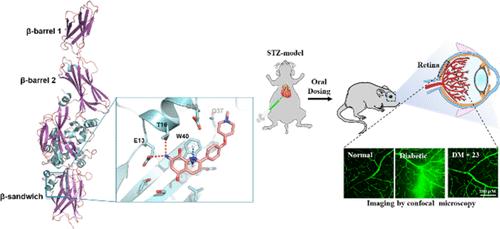
Diabetic retinopathy is a disease that can cause vision loss leading to blindness in people with diabetes. Improved methods to treat and prevent vision loss in diabetic patients are in high demand owing to limited current treatment procedures. Herein, we report a new class of transglutaminase 2 (TGase2) inhibitors for the treatment of diabetic retinopathy based on 7-aminoquinoline-5,8-dione derivatives. 7-Amino-2-phenylquinoline-5,8-dione 11 and 7-amino-2-{4-[(1-methylpiperidin-4-yl)oxy]phenyl}quinoline-5,8-dione 23 exhibited potent inhibitory activities against TGase2 in a fibrinogen array-based on-chip TGase2 activity assay and in an in situ assay in human retinal microvascular endothelial cells, with IC50 values of 5.88 and 1.12 μM in vitro, and 0.09 and 0.07 μM in situ, respectively. Pharmacokinetically favorable 7-amino-2-{4-[(1-isopropylpiperidin-4-yl)oxy] phenyl}quinoline-5,8-dione 22 inhibited vascular leakage in the retinas of streptozotocin-induced diabetic mice via oral administration. Results from the AL5 kinetic assay and a molecular docking study suggest that the inhibitors may bind to TGase2 remote from the active site.
用于治疗糖尿病视网膜病变的基于 7-氨基喹啉-5,8-二酮支架的结构最小化和药物样 TGase2 抑制剂
糖尿病视网膜病变是一种可导致糖尿病患者视力下降并最终失明的疾病。由于目前的治疗方法有限,因此需要改进治疗和预防糖尿病患者视力丧失的方法。在此,我们报告了一类基于 7-氨基喹啉-5,8-二酮衍生物的治疗糖尿病视网膜病变的新型转谷氨酰胺酶 2(TGase2)抑制剂。7-氨基-2-苯基喹啉-5,8-二酮 11 和 7-氨基-2-{4-[(1-甲基哌啶-4-基)氧基]苯基}喹啉-5,8-二酮 23 在基于纤维蛋白原阵列的片上 TGase2 活性测定和人视网膜微血管内皮细胞原位测定中表现出对 TGase2 的强效抑制活性,IC50 值分别为 5.体外的 IC50 值分别为 5.88 和 1.12 μM,原位的 IC50 值分别为 0.09 和 0.07 μM。药代动力学上有利的 7-氨基-2-{4-[(1-异丙基哌啶-4-基)氧基]苯基}喹啉-5,8-二酮 22 通过口服可抑制链脲佐菌素诱导的糖尿病小鼠视网膜的血管渗漏。AL5 动力学测定和分子对接研究的结果表明,这些抑制剂可能与远离活性位点的 TGase2 结合。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

Journal of Medicinal Chemistry
医学-医药化学
CiteScore
4.00
自引率
11.00%
发文量
804
审稿时长
1.9 months
期刊介绍:
The Journal of Medicinal Chemistry is a prestigious biweekly peer-reviewed publication that focuses on the multifaceted field of medicinal chemistry. Since its inception in 1959 as the Journal of Medicinal and Pharmaceutical Chemistry, it has evolved to become a cornerstone in the dissemination of research findings related to the design, synthesis, and development of therapeutic agents.
The Journal of Medicinal Chemistry is recognized for its significant impact in the scientific community, as evidenced by its 2022 impact factor of 7.3. This metric reflects the journal's influence and the importance of its content in shaping the future of drug discovery and development. The journal serves as a vital resource for chemists, pharmacologists, and other researchers interested in the molecular mechanisms of drug action and the optimization of therapeutic compounds.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


