Discovery of Potent and Selective Blockers Targeting the Epilepsy-Associated KNa1.1 Channel
IF 6.8
1区 医学
Q1 CHEMISTRY, MEDICINAL
引用次数: 0
Abstract
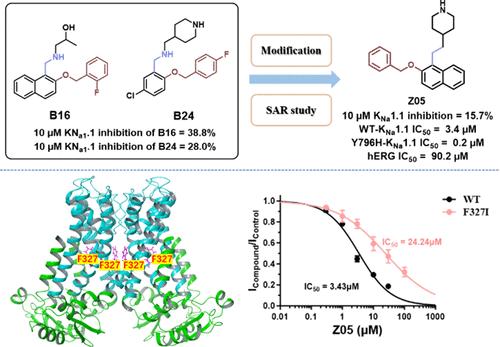
Gain-of-function (GOF) mutations of the sodium-activated potassium channel KNa1.1 (Slack, Slo2.2, or KCa4.1) induce severe, drug-resistant forms of epilepsy in infants and children. Although quinidine has shown promise in treating KCNT1-related epilepsies compared to other drugs, its limited efficacy and substantial side effects necessitate the development of new KNa1.1 channel inhibitors. In this study, we developed a novel class of KNa1.1 inhibitors using combined silico approaches and structural optimization. Among these inhibitors, compound Z05 was identified as a selective potential KNa1.1 inhibitor, especially against the hERG channel. Moreover, its binding site and potential counteraction to a GOF mutant Y796H were identified by the mutation studies. Our data also showed that Z05 had significant pharmacological profiles, including high brain penetration and moderate oral bioavailability, offering a valuable in vitro tool compound for further drug development in treating KCNT1-related epilepsies.
发现针对癫痫相关 KNa1.1 通道的强效选择性阻断剂
钠激活钾通道 KNa1.1(Slack、Slo2.2 或 KCa4.1)的功能增益(GOF)突变会诱发婴儿和儿童严重的抗药性癫痫。虽然与其他药物相比,奎尼丁在治疗 KCNT1 相关癫痫方面显示出了前景,但其有限的疗效和巨大的副作用使得开发新的 KNa1.1 通道抑制剂成为必要。在这项研究中,我们采用联合硅学方法和结构优化技术开发了一类新型 KNa1.1 抑制剂。在这些抑制剂中,化合物 Z05 被鉴定为一种潜在的 KNa1.1 选择性抑制剂,尤其是针对 hERG 通道。此外,通过突变研究还确定了它与 GOF 突变体 Y796H 的结合位点和潜在的反作用。我们的数据还显示,Z05 具有显著的药理学特征,包括高脑渗透性和适度的口服生物利用度,为进一步开发治疗 KCNT1 相关癫痫的药物提供了一种有价值的体外工具化合物。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

Journal of Medicinal Chemistry
医学-医药化学
CiteScore
4.00
自引率
11.00%
发文量
804
审稿时长
1.9 months
期刊介绍:
The Journal of Medicinal Chemistry is a prestigious biweekly peer-reviewed publication that focuses on the multifaceted field of medicinal chemistry. Since its inception in 1959 as the Journal of Medicinal and Pharmaceutical Chemistry, it has evolved to become a cornerstone in the dissemination of research findings related to the design, synthesis, and development of therapeutic agents.
The Journal of Medicinal Chemistry is recognized for its significant impact in the scientific community, as evidenced by its 2022 impact factor of 7.3. This metric reflects the journal's influence and the importance of its content in shaping the future of drug discovery and development. The journal serves as a vital resource for chemists, pharmacologists, and other researchers interested in the molecular mechanisms of drug action and the optimization of therapeutic compounds.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


