rGO/MUT-15 nanocomposite as a Fenton-like photocatalyst for the degradation of Acid Yellow 73 under visible light
IF 3.5
3区 化学
Q2 CHEMISTRY, INORGANIC & NUCLEAR
引用次数: 0
Abstract
The Fenton-like reaction is an advanced oxidation process (AOP) used to effectively eliminate organic pollutants. Fenton-like materials include metal–organic frameworks (MOFs) containing Fe, Co, Mn, and Cu metal ions. MOF-based photocatalysts with the highest performance can be designed and synthesized using these metal ions. A new Mn-based metal–organic framework with the formula of [Mn2(DClTPA)2(DMF)3] (MUT-15) containing 2,5-dichloroterephthalic acid (DClTPA) and N,N-dimethylformamide (DMF) was prepared via a solvothermal method. According to single-crystal X-ray analysis, MUT-15 (MUT = Materials from University of Tehran) has a tetragonal crystal system with the I41/a space group. A simple one-pot solvothermal method was used to prepare a rGO/MUT-15 nanocomposite. PXRD, FT-IR, TGA, FE-SEM, TEM, EDX, DRS, PL, EIS, and Mott–Schottky measurements were used to characterize the MUT-15 and rGO/MUT-15 nanocomposite. Under visible-light irradiation, MUT-15 and rGO/MUT-15 as Fenton-like photocatalysts degraded Acid Yellow 73 in only 10 min with outstanding photocatalytic activity rates of 92.39% and 96.10%, respectively. Thus, the Mn(II)–O clusters in MUT-15 significantly contributed to the degradation of Acid Yellow 73 through their Fenton-like effect.
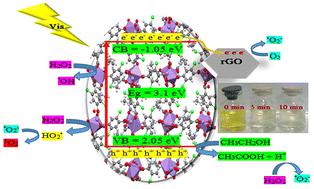
类芬顿反应是一种高级氧化过程(AOP),用于有效消除有机污染物。类芬顿材料包括含有铁、钴、锰和铜金属离子的金属有机框架(MOFs)。使用这些金属离子可以设计和合成具有最高性能的基于 MOF 的光催化剂。本研究通过溶热法制备了一种新的锰基金属有机框架,其分子式为[Mn2(DClTPA)2(DMF)3](MUT-15),含有 2,5-二氯对苯二甲酸(DClTPA)和 N,N-二甲基甲酰胺(DMF)。根据单晶 X 射线分析,MUT-15(MUT = 德黑兰大学材料)具有 I41/a 空间群的四方晶系。采用简单的一锅溶热法制备了 rGO/MUT-15 纳米复合材料。使用 PXRD、FT-IR、TGA、FE-SEM、TEM、EDX、DRS、PL、EIS 和 Mott-Schottky 测量法对 MUT-15 和 rGO/MUT-15 纳米复合材料进行了表征。在可见光照射下,MUT-15 和 rGO/MUT-15 作为 Fenton 类光催化剂仅在 10 分钟内就降解了酸性黄 73,光催化活性率分别达到 92.39% 和 96.10%。因此,MUT-15 中的 Mn(II)-O 团簇通过其 Fenton 类效应显著促进了酸性黄 73 的降解。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

Dalton Transactions
化学-无机化学与核化学
CiteScore
6.60
自引率
7.50%
发文量
1832
审稿时长
1.5 months
期刊介绍:
Dalton Transactions is a journal for all areas of inorganic chemistry, which encompasses the organometallic, bioinorganic and materials chemistry of the elements, with applications including synthesis, catalysis, energy conversion/storage, electrical devices and medicine. Dalton Transactions welcomes high-quality, original submissions in all of these areas and more, where the advancement of knowledge in inorganic chemistry is significant.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


