Anti-viral defence by an mRNA ADP-ribosyltransferase that blocks translation
IF 50.5
1区 综合性期刊
Q1 MULTIDISCIPLINARY SCIENCES
引用次数: 0
Abstract
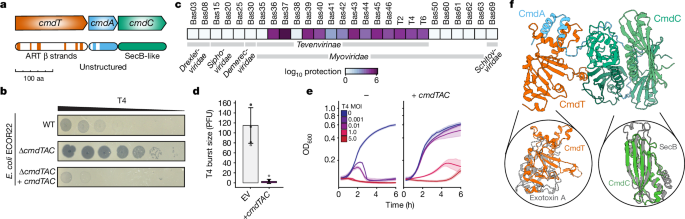
Host–pathogen conflicts are crucibles of molecular innovation1,2. Selection for immunity to pathogens has driven the evolution of sophisticated immunity mechanisms throughout biology, including in bacterial defence against bacteriophages3. Here we characterize the widely distributed anti-phage defence system CmdTAC, which provides robust defence against infection by the T-even family of phages4. Our results support a model in which CmdC detects infection by sensing viral capsid proteins, ultimately leading to the activation of a toxic ADP-ribosyltransferase effector protein, CmdT. We show that newly synthesized capsid protein triggers dissociation of the chaperone CmdC from the CmdTAC complex, leading to destabilization and degradation of the antitoxin CmdA, with consequent liberation of the CmdT ADP-ribosyltransferase. Notably, CmdT does not target a protein, DNA or structured RNA, the known targets of other ADP-ribosyltransferases. Instead, CmdT modifies the N6 position of adenine in GA dinucleotides within single-stranded RNAs, leading to arrest of mRNA translation and inhibition of viral replication. Our work reveals a novel mechanism of anti-viral defence and a previously unknown but broadly distributed class of ADP-ribosyltransferases that target mRNA. The bacterial anti-phage toxin–antitoxin–chaperone defence system CmdTAC senses capsid proteins via CmdC, enabling dissociation from the CmdTAC complex of the RNA ADP-ribosyltransferase CmdT, which targets single-stranded RNAs, inhibiting viral replication.

阻断翻译的 mRNA ADP-核糖基转移酶的抗病毒作用
宿主-病原体冲突是分子创新的关键1,2。对病原体免疫的选择推动了整个生物学中复杂免疫机制的进化,包括细菌对噬菌体的防御3。在这里,我们描述了广泛分布的抗噬菌体防御系统 CmdTAC 的特征,该系统能有效抵御噬菌体 T-even 家族的感染4。我们的研究结果支持这样一种模式,即 CmdC 通过感知病毒噬菌体蛋白来检测感染,最终导致毒性 ADP 核糖基转移酶效应蛋白 CmdT 的激活。我们的研究表明,新合成的噬菌体蛋白会触发伴侣 CmdC 从 CmdTAC 复合物中解离,导致抗毒素 CmdA 的不稳定和降解,从而释放出 CmdT ADP-核糖基转移酶。值得注意的是,CmdT 并不以蛋白质、DNA 或结构化 RNA 为靶标,而其他 ADP 核糖基转移酶的已知靶标是蛋白质、DNA 或结构化 RNA。相反,CmdT 会改变单链 RNA 中 GA 双核苷酸中腺嘌呤的 N6 位置,导致 mRNA 翻译停止并抑制病毒复制。我们的研究揭示了一种新的抗病毒防御机制,以及一种以前未知但分布广泛的以 mRNA 为靶标的 ADP 核糖基转移酶。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

Nature
综合性期刊-综合性期刊
CiteScore
90.00
自引率
1.20%
发文量
3652
审稿时长
3 months
期刊介绍:
Nature is a prestigious international journal that publishes peer-reviewed research in various scientific and technological fields. The selection of articles is based on criteria such as originality, importance, interdisciplinary relevance, timeliness, accessibility, elegance, and surprising conclusions. In addition to showcasing significant scientific advances, Nature delivers rapid, authoritative, insightful news, and interpretation of current and upcoming trends impacting science, scientists, and the broader public. The journal serves a dual purpose: firstly, to promptly share noteworthy scientific advances and foster discussions among scientists, and secondly, to ensure the swift dissemination of scientific results globally, emphasizing their significance for knowledge, culture, and daily life.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


