Modulating CO2 Electroreduction Activity on Mo2C and Promoting C2 Product by Grain Boundary Engineering: Insights from First-Principles Calculations
IF 11.3
1区 化学
Q1 CHEMISTRY, PHYSICAL
引用次数: 0
Abstract
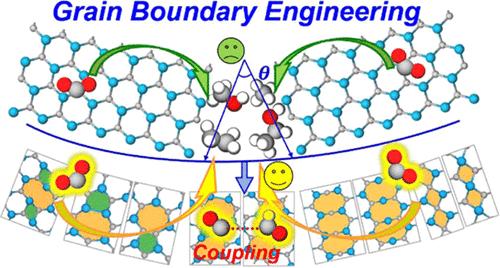
Recently, two-dimensional transition-metal carbides and/or nitrides (MXenes) have attracted extensive interest owing to their promising applications in electrochemistry, especially in electrocatalysis for the CO2 reduction reaction (CO2RR). However, there still exist challenges in developing MXene electrocatalysts with high activity and selectivity. Grain boundaries (GBs) could potentially provide active sites for chemical reactions, and their existence may be helpful for improving various electrocatalytic performances of MXenes. In this work, we constructed nine types of GBs in the Mo2C monolayer and employed density functional theory (DFT) calculations to systematically investigate their effects on the conversion efficiency of CO2 and the diversity of CO2RR products. Our study reveals that the presence of different valence states of Mo atoms at the GBs breaks the symmetry of CO2 adsorption on Mo2C, which promotes the activation of CO2 and diversifies the CO2RR products. Especially, these GBs exhibited remarkably low limiting potentials for C1 products, e.g., −0.29 V for CH4 on 5|7c GB, −0.31 V for CH3OH on 4|8 GB, and −0.55 V for HCOOH on 4|4a GB. Furthermore, the reduced potential barriers at the GBs, such as 0.26 eV for 5|7b GB and 0.13 eV for 8|8b GB, facilitate the C–C coupling and promote the formation of C2 products. These findings demonstrate that the introduction of GBs can enhance both the electrocatalytic activity of Mo2C for the CO2RR and the diversity of CO2RR products, therefore paving the way for designing and advancing high-efficiency MXene electrocatalysts through GB engineering.
求助全文
约1分钟内获得全文
求助全文
来源期刊

ACS Catalysis
CHEMISTRY, PHYSICAL-
CiteScore
20.80
自引率
6.20%
发文量
1253
审稿时长
1.5 months
期刊介绍:
ACS Catalysis is an esteemed journal that publishes original research in the fields of heterogeneous catalysis, molecular catalysis, and biocatalysis. It offers broad coverage across diverse areas such as life sciences, organometallics and synthesis, photochemistry and electrochemistry, drug discovery and synthesis, materials science, environmental protection, polymer discovery and synthesis, and energy and fuels.
The scope of the journal is to showcase innovative work in various aspects of catalysis. This includes new reactions and novel synthetic approaches utilizing known catalysts, the discovery or modification of new catalysts, elucidation of catalytic mechanisms through cutting-edge investigations, practical enhancements of existing processes, as well as conceptual advances in the field. Contributions to ACS Catalysis can encompass both experimental and theoretical research focused on catalytic molecules, macromolecules, and materials that exhibit catalytic turnover.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


