Decoding Hydrogen Desorption Kinetics in Porous Silicon: An Electrical Circuit Modeling Approach
IF 8.3
2区 材料科学
Q1 MATERIALS SCIENCE, MULTIDISCIPLINARY
引用次数: 0
Abstract
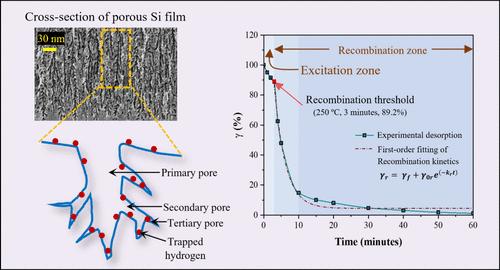
Solid-state hydrogen storage outperforms conventional storage methods in terms of safety and on-board applications. Porous Si (PS) is the optimized Si nanostructure with ample surface area (∼400 m2 g–1) and maximum dangling sites for hydrogenation. Though solid-state hydrogen storage in Si nanostructures, especially in porous Si, is extensively studied, the thermal desorption of hydrogen is rarely reported. This work investigates and analyzes the thermal desorption of a hydrogen-terminated PS surface using attenuated total reflectance–Fourier transform infrared spectroscopy (ATR-FTIR) to optimize the temperature for efficient desorption, as FTIR is sensitive to identifying the presence of Si hydride species (SiHx). The relative peak intensities in the spectra estimate the relative hydrogen retention (γ) for the analysis of the desorption kinetics. The desorption curves are divided into two zones on the time scale: the excitation zone and the recombination zone, separated by the recombination threshold point. The initially absorbed energy breaks the Si–Hx bonds in the excitation zone to reach the recombination threshold for H2 formation. The recombination zone is further divided into two subzones: the avalanche subzone (a sudden decrease in γ indicating molecular desorption) and the saturation subzone (almost constant γ with minimal desorption). The time constant from the first-order reaction kinetic fitting of the desorption curves explores the time–temperature correlation and the barrier energy estimation for the excitation and recombination zones. The analysis identifies the critical operating point for desorption as 100 °C and 4 min, with the optimized temperature of 250 °C. This article applies an analogous electrical circuit to compare the thermal hydrogen desorption and capacitor discharge circuit for analytical convenience.
解码多孔硅中的氢气解吸动力学:电路建模方法
固态储氢在安全性和车载应用方面优于传统储氢方法。多孔硅(PS)是一种优化的硅纳米结构,具有充足的表面积(∼400 m2 g-1)和最大的氢化悬垂位点。虽然人们对硅纳米结构,尤其是多孔硅中的固态储氢进行了广泛的研究,但氢的热解吸却鲜有报道。本研究利用衰减全反射-傅立叶变换红外光谱(ATR-FTIR)对氢封端 PS 表面的热解吸进行了研究和分析,以优化温度,实现高效解吸,因为傅立叶变换红外光谱能敏感地识别硅氢化物(SiHx)的存在。光谱中的相对峰强度可估算出相对氢保留度 (γ),用于分析解吸动力学。解吸曲线在时间尺度上分为两个区域:激发区和重组区,以重组阈值点为分界。最初吸收的能量会打断激发区中的 Si-Hx 键,从而达到 H2 形成的重组阈值。重组区又分为两个子区:雪崩子区(γ 突然降低,表明分子解吸)和饱和子区(γ 几乎恒定,解吸量极小)。从一阶反应动力学拟合解吸曲线得出的时间常数探索了激发区和重组区的时间-温度相关性和势垒能估算。分析确定解吸的临界工作点为 100 ℃ 和 4 分钟,优化温度为 250 ℃。为了便于分析,本文采用了类比电路来比较热解吸氢和电容器放电电路。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

ACS Applied Materials & Interfaces
工程技术-材料科学:综合
CiteScore
16.00
自引率
6.30%
发文量
4978
审稿时长
1.8 months
期刊介绍:
ACS Applied Materials & Interfaces is a leading interdisciplinary journal that brings together chemists, engineers, physicists, and biologists to explore the development and utilization of newly-discovered materials and interfacial processes for specific applications. Our journal has experienced remarkable growth since its establishment in 2009, both in terms of the number of articles published and the impact of the research showcased. We are proud to foster a truly global community, with the majority of published articles originating from outside the United States, reflecting the rapid growth of applied research worldwide.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


