Ferrocene Chalcone Enhanced Cyclotriphosphazene Photodiode Systems via Click Chemistry: Their Synthesis, Electrical, and Photophysical Properties
IF 4.3
2区 化学
Q1 CHEMISTRY, INORGANIC & NUCLEAR
引用次数: 0
Abstract
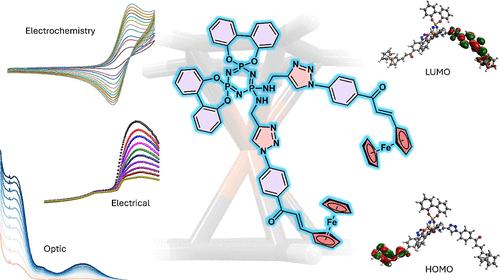
In this study, a new ferrocene-decorated cyclotriphosphazene compound (4) was obtained by the interaction of ferrocene chalcone with azide end groups (2) and spirophosphazene (3) bearing alkyne groups using the click method. The structures of all compounds were confirmed using Fourier transform infrared, 1H, 13C APT, and 31P NMR, and MS techniques. The optical band gaps of compounds were measured, showing values of 4.75 eV for 3, 3.53 eV for 2, and 3.46 eV for final product 4. The optical band gap of compound 4 calculated by density functional theory is 3.28 eV. The electrical properties of the compounds were investigated against the frequency. The results showed that the conduction mechanism is based on the hopping model. New types of diodes were prepared by using compounds 2 and 4. The current–voltage characteristics of the compound 4 diode showed good rectification behavior. The rectification ratio of the heterojunction diode was calculated as 629. The ideality factors were 3.35 for 2 and 7.57 for 4. The barrier heights were 0.68 and 0.92, respectively. Under sunlight (20–100 mW/cm2), both diodes exhibited photodiode properties, with compound 4 being more sensitive to light. This indicates that the diodes can be used in optoelectronic devices due to their photoconductive behavior.
求助全文
约1分钟内获得全文
求助全文
来源期刊

Inorganic Chemistry
化学-无机化学与核化学
CiteScore
7.60
自引率
13.00%
发文量
1960
审稿时长
1.9 months
期刊介绍:
Inorganic Chemistry publishes fundamental studies in all phases of inorganic chemistry. Coverage includes experimental and theoretical reports on quantitative studies of structure and thermodynamics, kinetics, mechanisms of inorganic reactions, bioinorganic chemistry, and relevant aspects of organometallic chemistry, solid-state phenomena, and chemical bonding theory. Emphasis is placed on the synthesis, structure, thermodynamics, reactivity, spectroscopy, and bonding properties of significant new and known compounds.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


