Manipulating Pt-ZnO interface for fast selective oxidation of glycerol to glyceric acid in base-free medium
IF 13.3
1区 工程技术
Q1 ENGINEERING, CHEMICAL
引用次数: 0
Abstract
Fine constructing the desirable active sites of catalysts is critical for the efficient polyol oxidation. Herein, we conceptually manipulate the Pt-ZnO interfacial active site instead of traditional Pt-Pt active site to isolate oxidation reaction and carboxylic acid desorption for fast selective oxidation of glycerol to glyceric acid in base-free medium. Multiple characterizations (including in-situ spectroscopy, reaction kinetics and density functional theory calculation, etc.) revealed that the strong electronic delocalization of Pt d-partial and the synergistic effect of ZnO adsorption could collectively strengthen the adsorption of glycerol and then promote the C-H bond of RCH2O intermediate in the primary hydroxyl group oxidation, resulting in higher catalytic activity over the Pt-ZnO interface. Meanwhile, the abundant oxygen vacancy of well-dispersed nano-ZnO could preferentially adsorb the formed glyceric acid product, preventing the coverage of the efficient Pt-Pt and Pt-O-Zn active sites by carboxylic acid products. Such tailored interface strategy achieves 80.4 % glycerol conversion and 84.6 % glyceric acid selectivity in only 1 h, and the turnover frequency could also be as high as 1338.4 h−1 over the optimal Pt/ZnO-MCM-41 (200) catalyst. The methodology provided here constitutes a promising basis for highly efficient polyol oxidation catalysis under mild and green conditions.
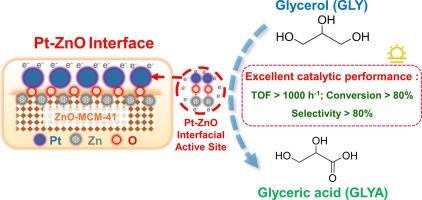
操纵铂-氧化锌界面,在无碱介质中将甘油快速选择性氧化为甘油酸
精细构建催化剂的理想活性位点对于高效多元醇氧化至关重要。在此,我们从概念上操纵 Pt-ZnO 界面活性位点而非传统的 Pt-Pt 活性位点,以隔离氧化反应和羧酸解吸,从而在无碱介质中将甘油快速选择性氧化为甘油酸。多种表征(包括原位光谱、反应动力学和密度泛函理论计算等)表明,铂的d-部分强电子分散和氧化锌的吸附协同效应可共同加强对甘油的吸附,进而促进初级羟基氧化过程中RCH2O中间体的C-H键,从而在Pt-ZnO界面上获得更高的催化活性。同时,分散良好的纳米 ZnO 中丰富的氧空位可以优先吸附形成的甘油酸产物,防止羧酸产物覆盖高效的 Pt-Pt 和 Pt-O-Zn 活性位点。这种量身定制的界面策略仅在 1 小时内就实现了 80.4% 的甘油转化率和 84.6% 的甘油酸选择性,在最佳的 Pt/ZnO-MCM-41 (200) 催化剂上,转化率还可高达 1338.4 h-1。本文提供的方法为在温和、绿色条件下进行高效多元醇氧化催化奠定了良好的基础。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

Chemical Engineering Journal
工程技术-工程:化工
CiteScore
21.70
自引率
9.30%
发文量
6781
审稿时长
2.4 months
期刊介绍:
The Chemical Engineering Journal is an international research journal that invites contributions of original and novel fundamental research. It aims to provide an international platform for presenting original fundamental research, interpretative reviews, and discussions on new developments in chemical engineering. The journal welcomes papers that describe novel theory and its practical application, as well as those that demonstrate the transfer of techniques from other disciplines. It also welcomes reports on carefully conducted experimental work that is soundly interpreted. The main focus of the journal is on original and rigorous research results that have broad significance. The Catalysis section within the Chemical Engineering Journal focuses specifically on Experimental and Theoretical studies in the fields of heterogeneous catalysis, molecular catalysis, and biocatalysis. These studies have industrial impact on various sectors such as chemicals, energy, materials, foods, healthcare, and environmental protection.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


