Cascade [3 + 2] Annulation of 1,3-Dicarbonyl Compounds and Ethyl Secondary Amines for Pyrrole Synthesis via Poly C(sp3)–H Bond Functionalization
IF 3.3
2区 化学
Q1 CHEMISTRY, ORGANIC
引用次数: 0
Abstract
The synthesis of polyfunctionalized pyrroles via the cascade reactions of 1,3-dicarbonyl compounds and two molecules of ethyl secondary amines has been realized via simple iodine catalysis in the presence of Dess–Martin periodinane (DMP). The formation of the target pyrrole products involves the formation of one new C–C and two new C–N bonds via the major functionalization of six C(sp3)–H bonds, presenting a highly novel and efficient synthetic protocol toward pyrrole scaffolds.
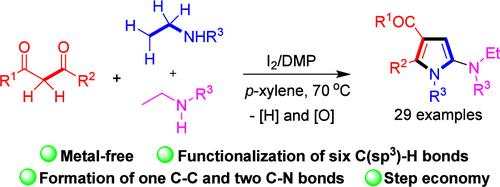
通过多 C(sp3)-H-键官能团化实现 1,3-二羰基化合物与乙基仲胺的级联 [3 + 2] 嵌合以合成吡咯
在 Dess-Martin periodinane(DMP)存在下,通过简单的碘催化反应,实现了 1,3-二羰基化合物和两分子乙基仲胺的级联反应,从而合成了多官能度的吡咯。目标吡咯产物的形成涉及通过六个 C(sp3)-H 键的主要官能化形成一个新的 C-C 键和两个新的 C-N 键,为吡咯支架的合成提供了一个高度新颖和高效的合成方案。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

The Journal of Organic Chemistry
化学-有机化学
CiteScore
6.20
自引率
11.10%
发文量
1467
审稿时长
2 months
期刊介绍:
The Journal of Organic Chemistry welcomes original contributions of fundamental research in all branches of the theory and practice of organic chemistry. In selecting manuscripts for publication, the editors place emphasis on the quality and novelty of the work, as well as the breadth of interest to the organic chemistry community.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


