Selective utilization of glucose metabolism guides mammalian gastrulation
IF 50.5
1区 综合性期刊
Q1 MULTIDISCIPLINARY SCIENCES
引用次数: 0
Abstract
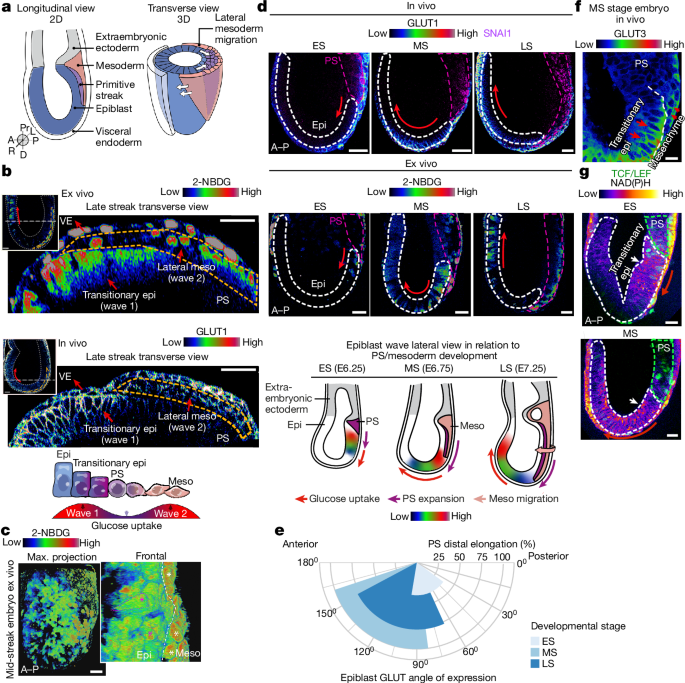
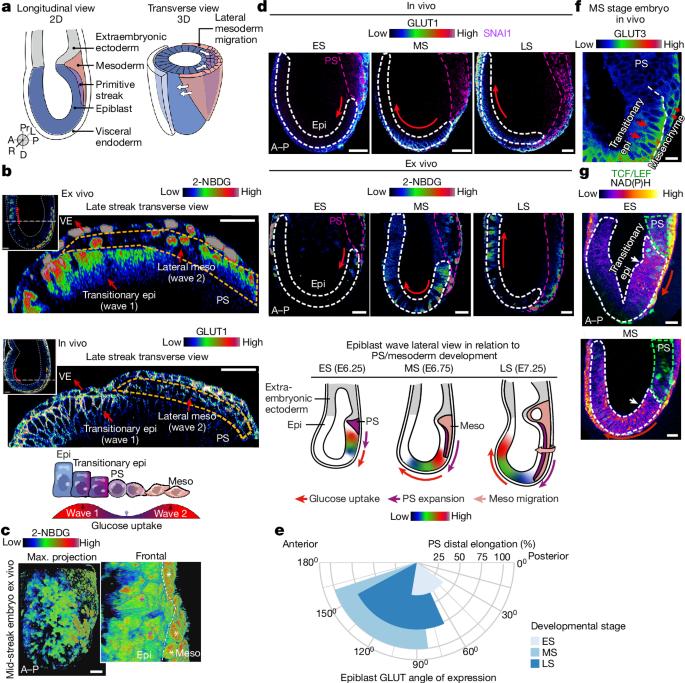
The prevailing dogma for morphological patterning in developing organisms argues that the combined inputs of transcription factor networks and signalling morphogens alone generate spatially and temporally distinct expression patterns. However, metabolism has also emerged as a critical developmental regulator1–10, independent of its functions in energy production and growth. The mechanistic role of nutrient utilization in instructing cellular programmes to shape the in vivo developing mammalian embryo remains unknown. Here we reveal two spatially resolved, cell-type- and stage-specific waves of glucose metabolism during mammalian gastrulation by using single-cell-resolution quantitative imaging of developing mouse embryos, stem cell models and embryo-derived tissue explants. We identify that the first spatiotemporal wave of glucose metabolism occurs through the hexosamine biosynthetic pathway to drive fate acquisition in the epiblast, and the second wave uses glycolysis to guide mesoderm migration and lateral expansion. Furthermore, we demonstrate that glucose exerts its influence on these developmental processes through cellular signalling pathways, with distinct mechanisms connecting glucose with the ERK activity in each wave. Our findings underscore that—in synergy with genetic mechanisms and morphogenic gradients—compartmentalized cellular metabolism is integral in guiding cell fate and specialized functions during development. This study challenges the view of the generic and housekeeping nature of cellular metabolism, offering valuable insights into its roles in various developmental contexts. Two waves of glucose metabolism provide distinct ERK-mediated cellular signals during gastrulation, which regulate cell fate and specialized cellular functions that are necessary for development.
葡萄糖代谢的选择性利用指导哺乳动物的胃形成
发育中生物体形态模式化的主流理论认为,仅转录因子网络和形态信号源的综合输入就能产生空间和时间上不同的表达模式。然而,新陈代谢也已成为一个关键的发育调节因子1,2,3,4,5,6,7,8,9,10,与其在能量生产和生长方面的功能无关。营养物质的利用在指导细胞程序以塑造体内发育中的哺乳动物胚胎方面的机制作用仍然未知。在这里,我们通过对发育中的小鼠胚胎、干细胞模型和胚胎衍生组织外植体进行单细胞分辨率定量成像,揭示了哺乳动物胚胎发育过程中葡萄糖代谢的两个空间分辨、细胞类型和阶段特异性波浪。我们发现,葡萄糖代谢的第一波时空波是通过己糖胺生物合成途径来驱动上胚层的命运获得,第二波是利用糖酵解来引导中胚层迁移和横向扩展。此外,我们还证明,葡萄糖通过细胞信号通路对这些发育过程施加影响,葡萄糖与每一波中的ERK活性之间都有不同的连接机制。我们的研究结果强调,在遗传机制和形态发生梯度的协同作用下,区系化细胞代谢在发育过程中引导细胞命运和特化功能不可或缺。这项研究挑战了细胞新陈代谢的一般和管家性质的观点,为了解细胞新陈代谢在各种发育环境中的作用提供了宝贵的见解。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

Nature
综合性期刊-综合性期刊
CiteScore
90.00
自引率
1.20%
发文量
3652
审稿时长
3 months
期刊介绍:
Nature is a prestigious international journal that publishes peer-reviewed research in various scientific and technological fields. The selection of articles is based on criteria such as originality, importance, interdisciplinary relevance, timeliness, accessibility, elegance, and surprising conclusions. In addition to showcasing significant scientific advances, Nature delivers rapid, authoritative, insightful news, and interpretation of current and upcoming trends impacting science, scientists, and the broader public. The journal serves a dual purpose: firstly, to promptly share noteworthy scientific advances and foster discussions among scientists, and secondly, to ensure the swift dissemination of scientific results globally, emphasizing their significance for knowledge, culture, and daily life.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


