Globally aromatic odd-electron π-magnetic macrocycle
IF 19.1
1区 化学
Q1 CHEMISTRY, MULTIDISCIPLINARY
引用次数: 0
Abstract
Molecular π-magnets based on single organic molecules have attracted increasing attention for their potential applications in optoelectronics and spintronics. Global aromaticity in conjugated macrocyclic polyradicaloids is still an open question that has only been tackled in molecules with an even number of electrons. Here, we report the on-surface synthesis of a cyclopenta-ring-fused oligo(m-phenylene) macrocycle, 9MC, with an odd number of electrons. The generated polyradicaloid undergoes a surface-induced distortion to a D3h symmetry with a fully delocalized doublet ground state. Interestingly, 9MC exhibits two aromatic annulene-within-an-annulene (AWA) ring currents in the inner and outer rings.
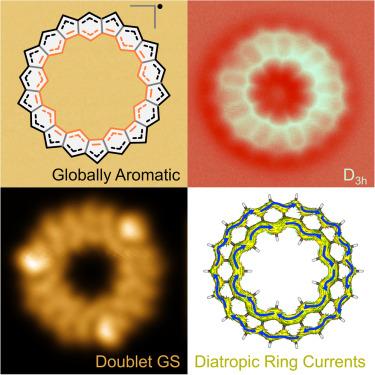

全芳香奇电子π-磁性大环
基于单一有机分子的分子π磁体因其在光电子学和自旋电子学中的潜在应用而受到越来越多的关注。共轭大环多核物的全局芳香性仍是一个未决问题,目前只在电子数为偶数的分子中得到解决。在此,我们报告了奇数电子数的环戊环融合低聚(间苯二酚)大环 9MC 的表面合成。生成的多辐射类化合物在表面诱导下畸变为 D3h 对称性,并具有完全去局域化的双基态。有趣的是,9MC 在内环和外环中显示出两个芳香环烯-环内-烯(AWA)环电流。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

Chem
Environmental Science-Environmental Chemistry
CiteScore
32.40
自引率
1.30%
发文量
281
期刊介绍:
Chem, affiliated with Cell as its sister journal, serves as a platform for groundbreaking research and illustrates how fundamental inquiries in chemistry and its related fields can contribute to addressing future global challenges. It was established in 2016, and is currently edited by Robert Eagling.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


