A validated high-performance liquid chromatography-ultraviolet method for the determination of valproic acid derivatives in pharmaceutical formulations with a microbiological suitability evaluation
IF 4.1
Q1 CHEMISTRY, ANALYTICAL
引用次数: 0
Abstract
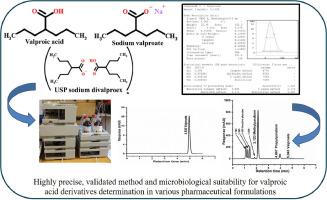
Valproic acid and its derivatives are common drugs used in the treatment of epilepsy, a result of CNS disorders. Because the molecular structure of valproic acid, as a fatty acid, does not contain any chromophore groups, its peak response demanded special conditions to be detected in HPLC, ensuring the best precision results. Still, the current approach illustrates simple, validated procedures for conduction. The conducted chromatographic system consists of the BDS Hypersil C8, 150 × 4.6 mm, 5 µm column using a mobile phase of acetonitrile: phosphate buffer (4:6) at a detection wavelength of 215 nm at room temperature. A full method validation study was conducted and approved to ensure precise, repeatable, and accurate results by implementing system suitability parameters. In microbiological terms, a suitability test is used to validate microbiological testing methods, ensuring their efficacy despite potential interference from antimicrobial properties in the tested materials. The HPLC-UV developed and validated analytical method was evaluated, and it was found to be sensitive for use in the detection of low concentrations of methylparaben, propylparaben, and valproate with an optimum run time of six minutes. limit of detection (LODs) were statistically estimated and found to be 2.27 µg/mL, 39.77 ng/mL, and 1.84 µg/mL for methylparaben, propylparaben, and valproate, respectively. Additionally, the method demonstrated a high recovery of methylparaben, propylparaben, and valproate with an excellent closed-accuracy range (98.84% - 101.34%). An excellent regression coefficient (r) of 0.99924, 0.99998, and 0.99997 for methylparaben, valproate, and propylparaben, respectively, was achieved. Assay determination of various pharmaceutical dosage forms in the local market was implemented, yielding admirable results.
高效液相色谱-紫外法测定药物制剂中丙戊酸衍生物的有效方法及微生物适用性评估
丙戊酸及其衍生物是治疗中枢神经系统疾病癫痫的常用药物。由于丙戊酸是一种脂肪酸,其分子结构中不含任何发色团,因此其峰值响应需要特殊条件才能在高效液相色谱法中检测到,以确保获得最佳精度结果。不过,目前的方法说明了简单、有效的传导程序。色谱系统采用 BDS Hypersil C8 色谱柱,150 × 4.6 mm,5 µm,流动相为乙腈:磷酸盐缓冲液(4:6),室温下检测波长为 215 nm。为确保检测结果的精确性、可重复性和准确性,我们进行了全面的方法验证研究,并通过了系统适用性参数的设置。从微生物学角度讲,适宜性测试用于验证微生物测试方法,确保这些方法在受测材料的抗菌特性可能造成干扰的情况下仍然有效。对开发和验证的 HPLC-UV 分析方法进行了评估,发现该方法灵敏度高,可用于检测低浓度的尼泊金甲酯、尼泊金丙酯和丙戊酸酯,最佳运行时间为 6 分钟。经统计估算,发现尼泊金甲酯、尼泊金丙酯和丙戊酸酯的检测限(LOD)分别为 2.27 µg/mL、39.77 ng/mL 和 1.84 µg/mL。此外,该方法还显示出对羟基苯甲酸甲酯、对羟基苯甲酸丙酯和丙戊酸钠的高回收率和极佳的闭合准确度范围(98.84% - 101.34%)。苯甲酸甲酯、丙戊酯和丙戊酯的回归系数(r)分别为 0.99924、0.99998 和 0.99997。对本地市场上的各种药物剂型进行了测定,结果令人满意。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文

 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


