Unlocking the stable interface in aqueous zinc-ion battery with multifunctional xylose-based electrolyte additives
IF 13.1
1区 化学
Q1 Energy
引用次数: 0
Abstract
The growth of dendrites and the side reactions occurring at the Zn anode pose significant challenges to the commercialization of aqueous Zn-ion batteries (AZIBs). These challenges arise from the inherent conflict between mass transfer and electrochemical kinetics. In this study, we propose the use of a multifunctional electrolyte additive based on the xylose (Xylo) molecule to address these issues by modulating the solvation structure and electrode/electrolyte interface, thereby stabilizing the Zn anode. The introduction of the additive alters the solvation structure, creating steric hindrance that impedes charge transfer and then reduces electrochemical kinetics. Furthermore, in-situ analyses demonstrate that the reconstructed electrode/electrolyte interface facilitates stable and rapid Zn2+ ion migration and suppresses corrosion and hydrogen evolution reactions. As a result, symmetric cells incorporating the Xylo additive exhibit significantly enhanced reversibility during the Zn plating/stripping process, with an impressively long lifespan of up to 1986 h, compared to cells using pure ZnSO4 electrolyte. When combined with a polyaniline cathode, the full cells demonstrate improved capacity and long-term cyclic stability. This work offers an effective direction for improving the stability of Zn anode via electrolyte design, as well as high-performance AZIBs.
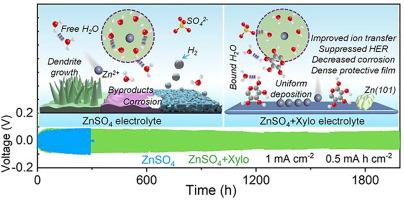
用基于木糖的多功能电解质添加剂打开锌离子水电池的稳定界面
枝晶的生长和锌阳极发生的副反应给水性锌离子电池(AZIB)的商业化带来了巨大挑战。这些挑战源于传质与电化学动力学之间的内在冲突。在本研究中,我们提出使用一种基于木糖(Xylo)分子的多功能电解质添加剂,通过调节溶解结构和电极/电解质界面来解决这些问题,从而稳定锌阳极。添加剂的引入改变了溶解结构,产生了立体阻碍,阻碍了电荷转移,进而降低了电化学动力学。此外,原位分析表明,重建的电极/电解质界面有利于 Zn2+ 离子稳定而快速地迁移,并抑制腐蚀和氢演化反应。因此,与使用纯 ZnSO4 电解液的电池相比,含有 Xylo 添加剂的对称电池在镀锌/剥离过程中的可逆性显著提高,使用寿命长达 1986 小时,令人印象深刻。当与聚苯胺阴极结合使用时,全电池的容量和长期循环稳定性都得到了提高。这项工作为通过电解质设计提高锌阳极的稳定性以及高性能 AZIB 提供了一个有效的方向。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

Journal of Energy Chemistry
CHEMISTRY, APPLIED-CHEMISTRY, PHYSICAL
CiteScore
19.10
自引率
8.40%
发文量
3631
审稿时长
15 days
期刊介绍:
The Journal of Energy Chemistry, the official publication of Science Press and the Dalian Institute of Chemical Physics, Chinese Academy of Sciences, serves as a platform for reporting creative research and innovative applications in energy chemistry. It mainly reports on creative researches and innovative applications of chemical conversions of fossil energy, carbon dioxide, electrochemical energy and hydrogen energy, as well as the conversions of biomass and solar energy related with chemical issues to promote academic exchanges in the field of energy chemistry and to accelerate the exploration, research and development of energy science and technologies.
This journal focuses on original research papers covering various topics within energy chemistry worldwide, including:
Optimized utilization of fossil energy
Hydrogen energy
Conversion and storage of electrochemical energy
Capture, storage, and chemical conversion of carbon dioxide
Materials and nanotechnologies for energy conversion and storage
Chemistry in biomass conversion
Chemistry in the utilization of solar energy
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


