Folic acid-functionalized and acetyl-terminated dendrimers as nanovectors for co-delivery of sorafenib and 5-fluorouracil
IF 3.8
3区 生物学
Q2 BIOCHEMISTRY & MOLECULAR BIOLOGY
引用次数: 0
Abstract
Molecular dynamics (MD) simulations were employed to investigate the simultaneous association of sorafenib (SF) and 5-fluorouracil (5-FU) with generation 4 (G4) acetyl-terminated poly(amidoamine) (PAMAM) dendrimers conjugated with folic acid (G4ACE-FA). Simulations were conducted under physiological (pH 7.4) and acidic (pH < 5) conditions, representing the environments of healthy and cancerous cells, respectively. The average radius of gyration (Rg) of G4ACE-FA was determined to be approximately 1.85 ± 0.01 nm and 2.31 ± 0.03 nm under physiological and acidic conditions, respectively. Drug loading did not exert a significant influence on the size and conformational compactness of G4ACE-FA at both neutral and low pH. However, a discernible increase in dendrimer size was observed upon simultaneous encapsulation and/or conjugation of both drug molecules. The relaxation times of G4ACE-FA were calculated to be 10.2 ns and 9.6 ns at neutral and low pH, respectively, indicating comparable equilibrium rates under both pH environments. The incorporation of small 5-FU molecules did not demonstrably alter the dendrimer's microstructure. The observed doubling of the relaxation time under acidic conditions can be attributed to the relatively compact structure of the dendrimer at neutral pH and the continuous intrastructural rearrangements occurring at acidic pH. The prolonged relaxation time observed in the G4ACE-FA:5-FU:SF complex is attributed to competitive interactions between 5-FU and SF molecules during simultaneous encapsulation by the dendrimer. Analysis of the unloaded and loaded structures of G4ACE-FA under varying pH conditions revealed a densely packed conformation at neutral pH and a more open, sponge-like structure at low pH. The solvent-accessible surface area (SASA) of the dendrimer was assessed at both pH conditions. At neutral pH, SASA values were approximately 124.0 ± 2.8 nm2, 127.5 ± 2.6 nm2, 131.3 ± 2.6 nm2, and 133.3 ± 2.6 nm2 for unloaded G4ACE-FA and the G4ACE-FA:5-FU, G4ACE-FA:SF, and G4ACE-FA:5-FU:SF complexes, respectively. Drug incorporation had a minimal effect on SASA at neutral pH. At low pH, the corresponding values were 198.2 ± 4.7 nm2, 195.8 ± 4.8 nm2, 212.5 ± 6.1 nm2, and 215.4 ± 4.2 nm2. These findings suggest that 5-FU encapsulation resulted in minimal changes to the dendrimer's surface exposure to the solvent, potentially due to its small size. In contrast, SF interaction led to a more pronounced increase in SASA, indicating structural expansion to accommodate SF conjugation. The equilibrium stoichiometry of the G4ACE-FA:5-FU complex was determined to be 1:11 and 1:3 at neutral and low pH, respectively. Similarly, the G4ACE-FA:SF complex exhibited equilibrium stoichiometries of 1:10 and 1:4 at neutral and low pH. The G4ACE-FA:5-FU:SF complex displayed stoichiometries of 1:11:10 at neutral pH and 1:3:3 at low pH. Collectively, these findings suggest that G4ACE-FA holds promise as a versatile nanovector capable of tightly binding drug molecules at neutral pH and facilitating their release within tumor cells, thereby enabling targeted drug delivery. Furthermore, the co-loading of 5-FU and SF did not compromise the loading capacity of G4ACE-FA. At neutral pH, 5-FU molecules were distributed evenly across the dendrimer surface and within its cavities, with 6 molecules encapsulated internally and 5 conjugated on the surface. At low pH, all bound 5-FU molecules were located at the dendrimer periphery. Similarly, at neutral pH, SF molecules were found both internally (6 molecules) and on the surface (4 molecules). At low pH, 2 SF molecules were found on the surface and 2 were internally complexed. The preferred binding sites of 5-FU and SF remained largely unchanged when co-loaded onto the dendrimer. This suggests that co-delivery of 5-FU and SF using G4ACE-FA could be a promising strategy for enhancing the therapeutic efficacy of these chemotherapeutic agents.
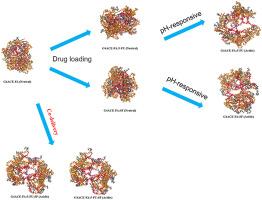
叶酸官能化和乙酰基端树枝状聚合物作为共同递送索拉非尼和 5-氟尿嘧啶的纳米载体。
我们采用分子动力学(MD)模拟研究了索拉非尼(SF)和5-氟尿嘧啶(5-FU)与叶酸共轭的第4代(G4)乙酰基端聚酰胺(PAMAM)树枝状聚合物(G4ACE-FA)同时结合的情况。模拟在生理(pH 值为 7.4)和酸性(pH 值小于 5)条件下进行,分别代表健康细胞和癌细胞的环境。在生理和酸性条件下,G4ACE-FA 的平均回旋半径 (Rg) 分别约为 1.85 ± 0.01 nm 和 2.31 ± 0.03 nm。在中性和低 pH 值条件下,药物负载对 G4ACE-FA 的大小和构象紧密性没有显著影响。不过,在同时包裹和/或共轭两种药物分子时,树枝状聚合物的尺寸明显增大。根据计算,G4ACE-FA 在中性和低 pH 值下的弛豫时间分别为 10.2 毫微秒和 9.6 毫微秒,这表明两种 pH 值环境下的平衡速率相当。5-FU 小分子的加入并未明显改变树枝状聚合物的微观结构。在酸性条件下观察到的弛豫时间加倍可归因于树枝状聚合物在中性 pH 值下相对紧凑的结构以及在酸性 pH 值下发生的持续的内部结构重排。在 G4ACE-FA:5-FU:SF 复合物中观察到的弛豫时间延长是由于 5-FU 和 SF 分子在同时被树枝状聚合物包裹的过程中发生了竞争性相互作用。对 G4ACE-FA 在不同 pH 值条件下的未负载和负载结构进行分析后发现,在中性 pH 值条件下,G4ACE-FA 呈致密堆积构象,而在低 pH 值条件下,G4ACE-FA 呈更开放的海绵状结构。在这两种 pH 值条件下,树枝状聚合物的溶剂可及表面积(SASA)都得到了评估。在中性 pH 值下,未负载 G4ACE-FA 和 G4ACE-FA:5-FU、G4ACE-FA:SF 和 G4ACE-FA:5-FU:SF 复合物的 SASA 值分别约为 124.0±2.8nm2、127.5±2.6nm2、131.3±2.6 nm2 和 133.3±2.6 nm2。在中性 pH 值下,药物掺入对 SASA 的影响很小。在低 pH 值时,相应的值分别为 198.2±4.7 nm2、195.8±4.8 nm2、212.5±6.1 nm2 和 215.4±4.2 nm2。这些研究结果表明,5-FU包封使树枝状聚合物暴露于溶剂的表面变化极小,这可能是由于树枝状聚合物体积小的缘故。与此相反,SF 的相互作用导致 SASA 的明显增加,这表明结构扩展以适应 SF 的共轭。在中性和低 pH 值条件下,G4ACE-FA:5-FU 复合物的平衡化学计量比分别为 1:11 和 1:3。同样,G4ACE-FA:SF 复合物在中性和低 pH 值条件下的平衡化学计量分别为 1:10 和 1:4。G4ACE-FA:5-FU:SF 复合物在中性 pH 值和低 pH 值条件下的平衡比例分别为 1:11:10和 1:3:3。总之,这些研究结果表明,G4ACE-FA有望成为一种多功能纳米载体,能够在中性pH值下紧密结合药物分子,并促进其在肿瘤细胞内的释放,从而实现靶向给药。此外,5-FU 和 SF 的共同负载并不影响 G4ACE-FA 的负载能力。在中性pH值下,5-FU分子均匀地分布在树枝状聚合物的表面和空腔内,其中6个分子被包裹在内部,5个分子共轭在表面。在低 pH 值时,所有结合的 5-FU 分子都位于树枝状聚合物的外围。同样,在中性 pH 值下,SF 分子既存在于内部(6 个分子),也存在于表面(4 个分子)。在低 pH 值条件下,2 个 SF 分子在表面,2 个在内部。当 5-FU 和 SF 共同负载到树枝状聚合物上时,它们的首选结合位点基本保持不变。这表明,使用 G4ACE-FA 共同递送 5-FU 和 SF 可能是提高这些化疗药物疗效的一种有前途的策略。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

Archives of biochemistry and biophysics
生物-生化与分子生物学
CiteScore
7.40
自引率
0.00%
发文量
245
审稿时长
26 days
期刊介绍:
Archives of Biochemistry and Biophysics publishes quality original articles and reviews in the developing areas of biochemistry and biophysics.
Research Areas Include:
• Enzyme and protein structure, function, regulation. Folding, turnover, and post-translational processing
• Biological oxidations, free radical reactions, redox signaling, oxygenases, P450 reactions
• Signal transduction, receptors, membrane transport, intracellular signals. Cellular and integrated metabolism.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


