Macrophages in the inflammatory response to endotoxic shock
Abstract
Background
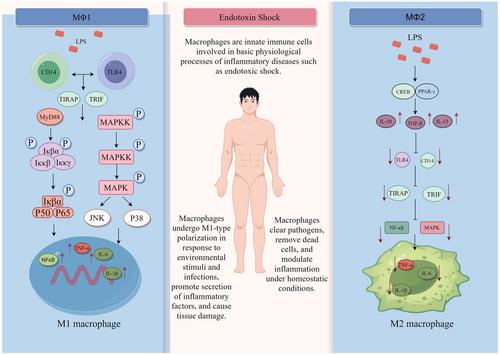
Endotoxic shock, particularly prevalent in intensive care units, represents a significant medical challenge. Endotoxin, upon invading the host, triggers intricate interactions with the innate immune system, particularly macrophages. This activation leads to the production of inflammatory mediators such as tumor necrosis factor-alpha, interleukin-6, and interleukin-1-beta, as well as aberrant activation of the nuclear factor-kappa-B and mitogen-activated protein kinase signaling pathways.
Objective
This review delves into the intricate inflammatory cascades underpinning endotoxic shock, with a particular focus on the pivotal role of macrophages. It aims to elucidate the clinical implications of these processes and offer insights into potential therapeutic strategies.
Results
Macrophages, central to immune regulation, manifest in two distinct subsets: M1 (classically activated subtype) macrophages and M2 (alternatively activated subtype) macrophages. The former exhibit an inflammatory phenotype, while the latter adopt an anti-inflammatory role. By modulating the inflammatory response in patients with endotoxic shock, these macrophages play a crucial role in restoring immune balance and facilitating recovery.
Conclusion
Macrophages undergo dynamic changes within the immune system, orchestrating essential processes for maintaining tissue homeostasis. A deeper comprehension of the mechanisms governing macrophage-mediated inflammation lays the groundwork for an anti-inflammatory, targeted approach to treating endotoxic shock. This understanding can significantly contribute to the development of more effective therapeutic interventions.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


