Polyvinylidene fluoride-based modified membranes for hydrogen generation by direct seawater electrolysis and proton exchange membrane fuel cells
IF 10.7
2区 材料科学
Q1 CHEMISTRY, PHYSICAL
引用次数: 0
Abstract
A polyvinylidene fluoride (PVDF) modified proton exchange membrane (PEM) bearing high sulfonic acid density was designed and investigated for water electrolysis application and H2–O2 fuel cell performance. The fabrication method involved ozonation of PVDF, followed by grafting using p-benzoquinone (i.e., Quino-PVDF) and successive sulfonation of Quino-PVDF to acquire the sulfonated Quino-PVDF copolymer. Moreover, a blending modification employing the Nafion™ ionomer was employed to enhance the performance of sulfonated Quino-PVDF based cation exchange membranes (i.e., QuinoCEMs). The membrane with 25 wt/wt% Nafion™/sulfonated Quino-PVDF (i.e., QuinoCEM-0.25) showed good performance in vapor-phase water electrolysis, liquid water electrolysis and direct seawater and achieved maximum current densities of 130, 480 and 240 mA cm−2 over a cell voltage of 1.8 V at 80 °C respectively. Furthermore, the QuinoCEM-0.25 based membrane electrode assembly achieved a peak power density of 400 mW cm−2, comparable to that of Nafion-212™ (i.e., 412 mW cm−2) in proton exchange membrane fuel cells (PEMFCs). Thus, this study highlights the potential of modified PVDF proton exchange membranes as efficient and cost-effective alternatives to commercially available PEMs.
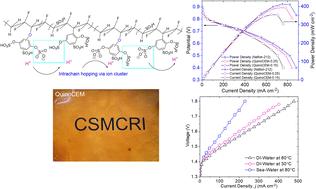
通过直接海水电解和质子交换膜燃料电池制氢的基于聚偏氟乙烯的改性膜
设计并研究了一种具有高磺酸密度的聚偏二氟乙烯(PVDF)改性质子交换膜(PEM),用于水电解应用和 H2-O2 燃料电池性能。制造方法包括对 PVDF 进行臭氧处理,然后使用对苯醌进行接枝(即 Quino-PVDF),并对 Quino-PVDF 进行连续磺化处理,以获得磺化 Quino-PVDF 共聚物。此外,还采用 Nafion™ 离子膜进行混合改性,以提高磺化 Quino-PVDF 阳离子交换膜(即 QuinoCEM)的性能。含 25 wt/wt% Nafion™/磺化 Quino-PVDF 的膜(即 QuinoCEM-0.25)在气相水电解、液态水电解和直接海水电解中均表现出良好的性能,在 80 °C 下,电池电压为 1.8 V 时的最大电流密度分别为 130、480 和 240 mA cm-2。此外,基于 QuinoCEM-0.25 的膜电极组件达到了 400 mW cm-2 的峰值功率密度,与质子交换膜燃料电池(PEMFC)中的 Nafion-212™(即 412 mW cm-2)相当。因此,本研究强调了改性 PVDF 质子交换膜作为高效、经济的商用 PEM 替代品的潜力。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

Journal of Materials Chemistry A
CHEMISTRY, PHYSICAL-ENERGY & FUELS
CiteScore
19.50
自引率
5.00%
发文量
1892
审稿时长
1.5 months
期刊介绍:
The Journal of Materials Chemistry A, B & C covers a wide range of high-quality studies in the field of materials chemistry, with each section focusing on specific applications of the materials studied. Journal of Materials Chemistry A emphasizes applications in energy and sustainability, including topics such as artificial photosynthesis, batteries, and fuel cells. Journal of Materials Chemistry B focuses on applications in biology and medicine, while Journal of Materials Chemistry C covers applications in optical, magnetic, and electronic devices. Example topic areas within the scope of Journal of Materials Chemistry A include catalysis, green/sustainable materials, sensors, and water treatment, among others.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


