Fabrication of Boron modified bi-functional air diffusion electrode for ciprofloxacin Degradation: In-situ H2O2 generation and self-activation
IF 13.3
1区 工程技术
Q1 ENGINEERING, CHEMICAL
引用次数: 0
Abstract
In order to improve H2O2 in-situ generation in electrochemical system, a boron (B) modified air diffusion electrode (B@ADE) was fabricated in the present study. Carbon black (CB) doped by B was deposited on ADE surface as catalytic layer. Performance of B@ADE with different B/C mass ratios has been investigated and 0.3 was identified as the optimal one·H2O2 yield can be achieved 204.85 mg L-1 within 45 min at 0.3-B@ADE, which was 1.51 times for that of ADE. Meanwhile, H2O2 self-activation on B@ADE surface was observed and effects of different operating parameters on •OH formation have been analyzed. XPS analyzation demonstrated that BC2O, BC2O and C = O/O-C = O were related to H2O2 generation. DFT calculation revealed that BCO2 was benefit for O2 adsorption, while BC2O promoted *OOH desorption with lower free energy barriers for H2O2 and •OH generation. Quenching experiments for ciprofloxacin (CIP) demonstrated that •OH, O•-2 and 1O2 were co-existed in the system with their contribution quantified. Four possible degradation pathways were proposed through active sites identification by DFT calculation and intermediates detection by HPLC-MS/MS. Bio-toxicity analyzation results showed that the toxicity of most intermediates was lowered than CIP. These results proved that electrochemical oxidation system with B@ADE enhanced in-situ H2O2 generation and self-activation, with great potential for practical application.
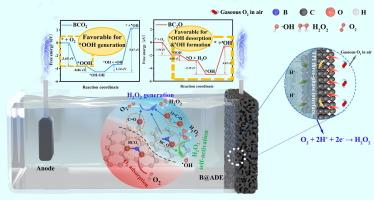
制造用于降解环丙沙星的硼修饰双功能空气扩散电极:原位 H2O2 生成与自激活
为了改善电化学系统中 H2O2 的原位生成,本研究制作了硼(B)修饰的空气扩散电极(B@ADE)。掺杂硼的炭黑(CB)沉积在 ADE 表面作为催化层。研究了不同 B/C 质量比的 B@ADE 的性能,发现 0.3 是最佳的 B@ADE 质量比,0.3-B@ADE 在 45 分钟内的 H2O2 产率可达 204.85 mg L-1,是 ADE 的 1.51 倍。同时,观察了 H2O2 在 B@ADE 表面的自活化情况,并分析了不同操作参数对 -OH 形成的影响。XPS 分析表明,BC2O、BC2O 和 C = O/O-C = O 与 H2O2 的生成有关。DFT 计算显示,BCO2 有利于 O2 的吸附,而 BC2O 则促进了 *OOH 的解吸,其生成 H2O2 和 -OH 的自由能垒较低。环丙沙星(CIP)的淬灭实验表明,-OH、O-2 和 1O2 在系统中共存,并对它们的贡献进行了量化。通过 DFT 计算确定活性位点和 HPLC-MS/MS 检测中间产物,提出了四种可能的降解途径。生物毒性分析结果表明,大多数中间产物的毒性低于 CIP。这些结果证明,使用 B@ADE 的电化学氧化系统增强了原位 H2O2 生成和自活化能力,具有巨大的实际应用潜力。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

Chemical Engineering Journal
工程技术-工程:化工
CiteScore
21.70
自引率
9.30%
发文量
6781
审稿时长
2.4 months
期刊介绍:
The Chemical Engineering Journal is an international research journal that invites contributions of original and novel fundamental research. It aims to provide an international platform for presenting original fundamental research, interpretative reviews, and discussions on new developments in chemical engineering. The journal welcomes papers that describe novel theory and its practical application, as well as those that demonstrate the transfer of techniques from other disciplines. It also welcomes reports on carefully conducted experimental work that is soundly interpreted. The main focus of the journal is on original and rigorous research results that have broad significance. The Catalysis section within the Chemical Engineering Journal focuses specifically on Experimental and Theoretical studies in the fields of heterogeneous catalysis, molecular catalysis, and biocatalysis. These studies have industrial impact on various sectors such as chemicals, energy, materials, foods, healthcare, and environmental protection.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


