Type I IFN drives unconventional IL-1β secretion in lupus monocytes
IF 26.3
1区 医学
Q1 IMMUNOLOGY
引用次数: 0
Abstract
Opsonization of red blood cells that retain mitochondria (Mito+ RBCs), a feature of systemic lupus erythematosus (SLE), triggers type I interferon (IFN) production in macrophages. We report that monocytes (Mos) co-produce IFN and mature interleukin-1β (mIL-1β) upon Mito+ RBC opsonization. IFN expression depended on cyclic GMP-AMP synthase (cGAS) and RIG-I-like receptors’ (RLRs) sensing of Mito+ RBC-derived mitochondrial DNA (mtDNA) and mtRNA, respectively. Interleukin-1β (IL-1β) production was initiated by the RLR antiviral signaling adaptor (MAVS) pathway recognition of Mito+ RBC-derived mtRNA. This led to the cytosolic release of Mo mtDNA, which activated the inflammasome. Importantly, mIL-1β secretion was independent of gasdermin D (GSDMD) and pyroptosis but relied on IFN-inducible myxovirus-resistant protein 1 (MxA), which facilitated the incorporation of mIL-1β into a trans-Golgi network (TGN)-mediated secretory pathway. RBC internalization identified a subset of blood Mo expressing IFN-stimulated genes (ISGs) that released mIL-1β and expanded in SLE patients with active disease.
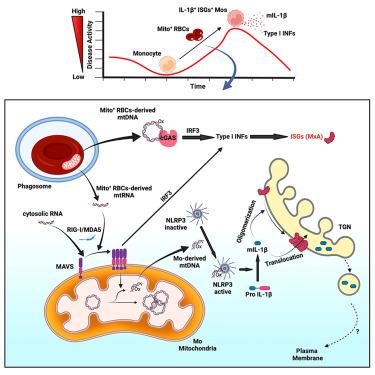
I 型 IFN 驱动狼疮单核细胞分泌非常规 IL-1β
保留线粒体的红细胞(线粒体+红细胞)被溶解是系统性红斑狼疮(SLE)的一个特征,它会触发巨噬细胞产生 I 型干扰素(IFN)。我们报告说,单核细胞(Mos)在Mito+ RBC蛋白溶解后会同时产生IFN和成熟的白细胞介素-1β(mIL-1β)。IFN 的表达分别取决于环 GMP-AMP 合成酶(cGAS)和类 RIG-I 受体(RLRs)对 Mito+ RBC 衍生的线粒体 DNA(mtDNA)和 mtRNA 的感应。白细胞介素-1β(IL-1β)的产生是由 RLR 抗病毒信号适配器(MAVS)途径识别 Mito+ RBC 衍生的 mtRNA 引发的。这导致细胞膜释放 Mo mtDNA,从而激活炎性体。重要的是,mIL-1β的分泌不依赖于gasdermin D(GSDMD)和化脓作用,而是依赖于IFN诱导的肌瘤病毒抗性蛋白1(MxA),MxA促进了mIL-1β进入由跨高尔基体网络(TGN)介导的分泌途径。红细胞内化确定了表达 IFN 刺激基因(ISGs)的血摩亚群,这些基因释放 mIL-1β,并在活动性系统性红斑狼疮患者中扩大。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

Immunity
医学-免疫学
CiteScore
49.40
自引率
2.20%
发文量
205
审稿时长
6 months
期刊介绍:
Immunity is a publication that focuses on publishing significant advancements in research related to immunology. We encourage the submission of studies that offer groundbreaking immunological discoveries, whether at the molecular, cellular, or whole organism level. Topics of interest encompass a wide range, such as cancer, infectious diseases, neuroimmunology, autoimmune diseases, allergies, mucosal immunity, metabolic diseases, and homeostasis.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


