Integrating strategy to investigate the formation and stabilization mechanisms of Curcumin-Cyclodextrin inclusion complexes: Experimental characterizations and multi-scale computational simulations
IF 11
1区 农林科学
Q1 CHEMISTRY, APPLIED
引用次数: 0
Abstract
The Cyclodextrin (CD) encapsulation technology can significantly enhance the water solubility of Curcumin (CUR) and effectively protect it against chemical degradations. In the current study, a combination of experimental and multi-scale computational approaches was used to investigate the binding mechanism between CUR and β-CDs (β-CD, 2-HP-β-CD, Me-β-CD, and DM-β-CD). Phase solubility studies showed that β-CDs exhibited a strong binding affinity with CUR and significantly enhanced its solubility. SEM, PXRD, DSC, and TG analysis confirmed the formation of inclusion complexes, resulting in the transition of CUR from crystalline to an amorphous state. FTIR, 1H, and 13C NMR spectra deduced that CUR may have multiple binding conformations with β-CDs. The rationality of molecular dynamics simulation systems was confirmed by comparing the simulated IR spectrum from quantum mechanics with the experimental IR spectrum. MM-PBSA and umbrella sampling simulation calculated the binding free energy of the CUR/CD inclusion complexes, ranking Me-β-CD > DM-β-CD > 2-HP-β-CD > β-CD, and clarified that van der Waals interaction played a major role in stabilizing the inclusion complexes. RDF analysis confirmed that the release of high-energy water molecules drove the formation of the CUR/CD inclusion complex, and the β-CD substituents affected their exterior hydration layer. Additionally, principal component analysis (PCA) and non-covalent interaction analysis revealed three CUR/CD binding modes: bead-on-string, terminal insertion, and V-shaped-folding. The research strategy adopted here can serve as a paradigm for investigating the encapsulation mechanism of poorly soluble drugs with CDs.
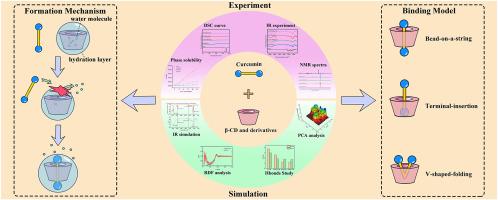
研究姜黄素-环糊精包合物的形成和稳定机制的综合策略:实验表征与多尺度计算模拟
环糊精(CD)包封技术可显著提高姜黄素(CUR)的水溶性,并有效防止其化学降解。本研究采用实验和多尺度计算相结合的方法研究了 CUR 与 β-CD(β-CD、2-HP-β-CD、Me-β-CD 和 DM-β-CD)之间的结合机制。相溶解度研究表明,β-CD 与 CUR 具有很强的结合亲和力,能显著提高 CUR 的溶解度。SEM、PXRD、DSC 和 TG 分析证实了包合物的形成,导致 CUR 从结晶状态转变为无定形状态。傅立叶变换红外光谱、1H 和 13C NMR 光谱推断出 CUR 与 β-CD 可能有多种结合构象。通过比较量子力学模拟红外光谱和实验红外光谱,证实了分子动力学模拟系统的合理性。MM-PBSA和伞状采样模拟计算了CUR/CD包合物的结合自由能,将Me-β-CD > DM-β-CD > 2-HP-β-CD > β-CD进行了排序,明确了范德华相互作用在稳定包合物中发挥了重要作用。RDF 分析证实,高能水分子的释放推动了 CUR/CD 包合物的形成,β-CD 取代基影响了其外部水合层。此外,主成分分析(PCA)和非共价相互作用分析揭示了三种 CUR/CD 结合模式:串珠结合、末端插入结合和 V 形折叠结合。本文采用的研究策略可作为研究 CD 对难溶性药物包封机理的范例。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

Food Hydrocolloids
工程技术-食品科技
CiteScore
19.90
自引率
14.00%
发文量
871
审稿时长
37 days
期刊介绍:
Food Hydrocolloids publishes original and innovative research focused on the characterization, functional properties, and applications of hydrocolloid materials used in food products. These hydrocolloids, defined as polysaccharides and proteins of commercial importance, are added to control aspects such as texture, stability, rheology, and sensory properties. The research's primary emphasis should be on the hydrocolloids themselves, with thorough descriptions of their source, nature, and physicochemical characteristics. Manuscripts are expected to clearly outline specific aims and objectives, include a fundamental discussion of research findings at the molecular level, and address the significance of the results. Studies on hydrocolloids in complex formulations should concentrate on their overall properties and mechanisms of action, while simple formulation development studies may not be considered for publication.
The main areas of interest are:
-Chemical and physicochemical characterisation
Thermal properties including glass transitions and conformational changes-
Rheological properties including viscosity, viscoelastic properties and gelation behaviour-
The influence on organoleptic properties-
Interfacial properties including stabilisation of dispersions, emulsions and foams-
Film forming properties with application to edible films and active packaging-
Encapsulation and controlled release of active compounds-
The influence on health including their role as dietary fibre-
Manipulation of hydrocolloid structure and functionality through chemical, biochemical and physical processes-
New hydrocolloids and hydrocolloid sources of commercial potential.
The Journal also publishes Review articles that provide an overview of the latest developments in topics of specific interest to researchers in this field of activity.
文献相关原料
公司名称
产品信息
上海源叶
Curcumin
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


