Discovery of BAY-405: An Azaindole-Based MAP4K1 Inhibitor for the Enhancement of T-Cell Immunity against Cancer
IF 6.8
1区 医学
Q1 CHEMISTRY, MEDICINAL
引用次数: 0
Abstract
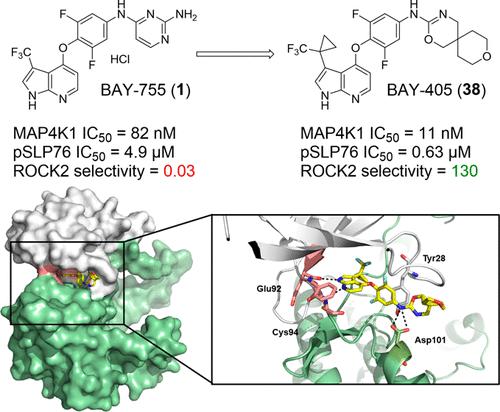
Mitogen-activated protein kinase kinase kinase kinase 1 (MAP4K1) is a serine/threonine kinase that acts as an immune checkpoint downstream of T-cell receptor stimulation. MAP4K1 activity is enhanced by prostaglandin E2 (PGE2) and transforming growth factor beta (TGFβ), immune modulators commonly present in the tumor microenvironment. Therefore, its pharmacological inhibition is an attractive immuno-oncology concept for inducing therapeutic T-cell responses in cancer patients. Here, we describe the systematic optimization of azaindole-based lead compound 1, resulting in the discovery of potent and selective MAP4K1 inhibitor 38 (BAY-405) that displays nanomolar potency in biochemical and cellular assays as well as in vivo exposure after oral dosing. BAY-405 enhances T-cell immunity and overcomes the suppressive effect of PGE2 and TGFβ. Treatment of tumor-bearing mice shows T-cell-dependent antitumor efficacy. MAP4K1 inhibition in conjunction with PD-L1 blockade results in a superior antitumor impact, illustrating the complementarity of the single agent treatments.
发现 BAY-405:一种基于氮杂吲哚的 MAP4K1 抑制剂,可增强 T 细胞的抗癌免疫力
丝氨酸/苏氨酸激酶 1(MAP4K1)是一种丝氨酸/苏氨酸激酶,在 T 细胞受体刺激的下游起着免疫检查点的作用。前列腺素 E2(PGE2)和转化生长因子β(TGFβ)是肿瘤微环境中常见的免疫调节剂,它们会增强 MAP4K1 的活性。因此,药理抑制前列腺素 E2 是诱导癌症患者治疗性 T 细胞应答的一个极具吸引力的免疫肿瘤学概念。在此,我们介绍了对氮杂环吲哚类先导化合物 1 的系统优化,从而发现了强效、选择性的 MAP4K1 抑制剂 38(BAY-405),它在生化和细胞实验中以及口服后的体内暴露中都显示出纳摩尔级的效力。BAY-405 可增强 T 细胞免疫力,克服 PGE2 和 TGFβ 的抑制作用。对肿瘤小鼠的治疗显示出T细胞依赖性抗肿瘤疗效。在抑制 MAP4K1 的同时阻断 PD-L1 会产生更好的抗肿瘤效果,这说明了单药治疗的互补性。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

Journal of Medicinal Chemistry
医学-医药化学
CiteScore
4.00
自引率
11.00%
发文量
804
审稿时长
1.9 months
期刊介绍:
The Journal of Medicinal Chemistry is a prestigious biweekly peer-reviewed publication that focuses on the multifaceted field of medicinal chemistry. Since its inception in 1959 as the Journal of Medicinal and Pharmaceutical Chemistry, it has evolved to become a cornerstone in the dissemination of research findings related to the design, synthesis, and development of therapeutic agents.
The Journal of Medicinal Chemistry is recognized for its significant impact in the scientific community, as evidenced by its 2022 impact factor of 7.3. This metric reflects the journal's influence and the importance of its content in shaping the future of drug discovery and development. The journal serves as a vital resource for chemists, pharmacologists, and other researchers interested in the molecular mechanisms of drug action and the optimization of therapeutic compounds.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


