CuI-Catalyzed Selenylation of Pyrrolo[2,1-a]isoquinolines and Other Electron-Rich Heteroarenes
IF 3.3
2区 化学
Q1 CHEMISTRY, ORGANIC
引用次数: 0
Abstract
We have established a mild CuI-catalyzed selenylation of pyrrolo[2,1-a]isoquinoline derivatives in the presence of mCPBA (m-chloroperoxybenzoic acid) at ambient temperature. Corresponding organoselenides have been prepared readily in 53–92% yields. This process can also be expanded to the modification of pyrroles, azaindole, and indoles, delivering the desired heterocyclic selenides in moderate to good yields.
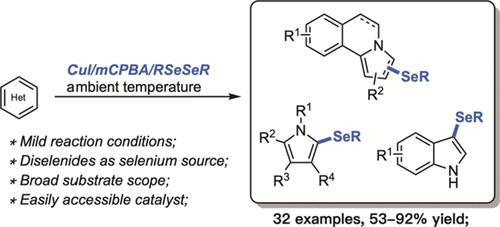
CuI 催化的吡咯并[2,1-a]异喹啉和其他富电子杂环戊烯的硒化反应
我们建立了一种在 mCPBA(间氯过氧苯甲酸)存在下,于室温对吡咯并[2,1-a]异喹啉衍生物进行温和的 CuI 催化硒化反应的方法。相应的有机硒化物很容易制备,产率为 53-92%。这一工艺还可扩展到吡咯、氮杂吲哚和吲哚的改性,以中等至良好的收率制备出所需的杂环硒化物。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

The Journal of Organic Chemistry
化学-有机化学
CiteScore
6.20
自引率
11.10%
发文量
1467
审稿时长
2 months
期刊介绍:
The Journal of Organic Chemistry welcomes original contributions of fundamental research in all branches of the theory and practice of organic chemistry. In selecting manuscripts for publication, the editors place emphasis on the quality and novelty of the work, as well as the breadth of interest to the organic chemistry community.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


