Grain boundary defect passivation and iodine migration inhibition for efficient and stable perovskite solar cells
IF 5.5
3区 材料科学
Q1 ELECTROCHEMISTRY
引用次数: 0
Abstract
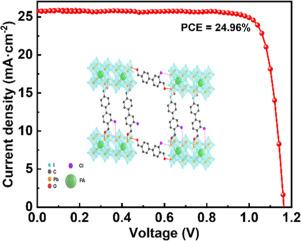
To inhibit the I−migration and passivate the uncoordinated Pb2+ on perovskite grain boundary (GB), 3-methyl-(1,1′-biphenyl)-4,4′-diformaldehyde (BPDA-Me) and 3-chlorine-(1,1′-biphenyl)-4,4′-diformaldehyde (BPDA-Cl) were synthesized and incorporated into perovskite films, respectively. The C = O groups in both additives can strongly interact with the uncoordinated Pb2+, allowing them to be anchored in perovskite GB. At the same time, the benzene ring skeleton in these two molecular structures can interact with the migrating I− in GB. The Cl atom in the BPDA-Cl molecule delocalize electrons into the C = O group due to the conjugation effect of the benzene ring, so that the C = O group can provide stronger electronegativity, thus enhancing the interaction with the uncoordinated Pb2+. Meanwhile, Cl atom can coordinate with Pb2+ to synergically passivate the I vacancy defect on GB and enhance the lattice strength of PbI6. This cooperative passivation further effectively inhibited the I−migration occurring at the perovskite GB. The functional group electron density regulation and cooperative passivation of Cl and C = O in BPDA-Cl make the passivation effect better than BPDA-Me. Consequently, the BPDA-Cl based perovskite solar cell achieved the highest power conversion efficiency of 24.96 % and stability with 92.1 % of the initial performance retained after 500 h of operation under continuous lighting and maximum power point tracking conditions.
晶界缺陷钝化和碘迁移抑制,实现高效稳定的过氧化物太阳能电池
为了抑制I迁移并钝化包晶晶界(GB)上未配位的Pb2+,合成了3-甲基-(1,1′-联苯)-4,4′-二甲醛(BPDA-Me)和3-氯-(1,1′-联苯)-4,4′-二甲醛(BPDA-Cl),并分别将其加入到包晶薄膜中。这两种添加剂中的 C = O 基团都能与未配位的 Pb2+ 发生强烈的相互作用,使它们能够锚定在包晶 GB 中。同时,这两种分子结构中的苯环骨架还能与 GB 中迁移的 I- 相互作用。由于苯环的共轭效应,BPDA-Cl 分子中的 Cl 原子会将电子析出到 C = O 基团中,这样 C = O 基团就能提供更强的电负性,从而增强与未配位 Pb2+ 的相互作用。同时,Cl 原子能与 Pb2+ 配位,协同钝化 GB 上的 I 空位缺陷,增强 PbI6 的晶格强度。这种协同钝化作用进一步有效地抑制了包晶 GB 上发生的 I 迁移。BPDA-Cl 中的官能团电子密度调节和 Cl 与 C = O 的协同钝化使其钝化效果优于 BPDA-Me。因此,在连续照明和最大功率点跟踪条件下,基于 BPDA-Cl 的过氧化物太阳能电池实现了最高的功率转换效率(24.96%)和稳定性,在运行 500 小时后,其初始性能仍能保持 92.1%。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

Electrochimica Acta
工程技术-电化学
CiteScore
11.30
自引率
6.10%
发文量
1634
审稿时长
41 days
期刊介绍:
Electrochimica Acta is an international journal. It is intended for the publication of both original work and reviews in the field of electrochemistry. Electrochemistry should be interpreted to mean any of the research fields covered by the Divisions of the International Society of Electrochemistry listed below, as well as emerging scientific domains covered by ISE New Topics Committee.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


