Lifecycle of Pd Clusters: Following the Formation and Evolution of Active Pd Clusters on Ceria During CO Oxidation by In Situ/Operando Characterization Techniques
IF 11.3
1区 化学
Q1 CHEMISTRY, PHYSICAL
引用次数: 0
Abstract
For maximizing the atomic efficiency in noble metal-based catalysts, dedicated preparation routes and high lifetime are essential. Both aspects require an in-depth understanding of the fate of noble metal atoms under reaction conditions. For this purpose, we used a combination of complementary in situ/operando characterization techniques to follow the lifecycle of the Pd sites in a 0.5% Pd/5% CeO2–Al2O3 catalyst during oxygen-rich CO oxidation. Time-resolved X-ray absorption spectroscopy showed that Pd cluster formation under reaction conditions is important for a high CO oxidation activity. In combination with density functional theory calculations, we concluded that the ideal Pd cluster size amounts to about 10–30 Pd atoms. The cluster formation and stability were affected by the applied temperature and reaction conditions. Already short pulses of 1000 ppm CO in the lean reaction feed were found to trigger sintering of Pd at temperatures below 200 °C, while at higher temperatures oxidation processes prevailed. Environmental transmission electron microscopy unraveled redispersion at higher temperatures (400–500 °C) in oxygen atmosphere, leading to the formation of single sites and thus the loss of activity. However, due to the reductive nature of CO, clusters formed again upon cooling in reaction atmosphere, thus closing the catalytic cycle. Exploiting the gained knowledge on the lifecycle of Pd clusters, we systematically investigated the effect of catalyst composition on the cluster formation tendency. As uncovered by DRIFTS measurements, the Pd to CeO2 ratio seems to be a key descriptor for Pd agglomeration under reaction conditions. While for higher Pd loadings, the probability of cluster formation increased, a higher CeO2 content leads to the formation of oxidized dispersed Pd species. According to our results, a Pd:CeO2 weight ratio of 1:10 for CeO2–Al2O3-supported catalysts leads to the highest CO oxidation activity under lean conditions independent of the applied synthesis method.
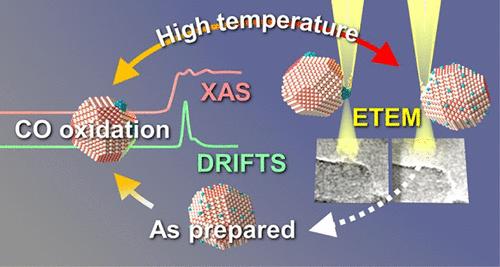
钯簇的生命周期:通过原位/操作表征技术跟踪一氧化碳氧化过程中铈上活性钯簇的形成和演化过程
要最大限度地提高贵金属基催化剂的原子效率,专用的制备路线和高寿命至关重要。这两方面都需要深入了解贵金属原子在反应条件下的去向。为此,我们结合使用了互补的原位/操作表征技术,以跟踪富氧 CO 氧化过程中 0.5% Pd/5% CeO2-Al2O3 催化剂中 Pd 位点的生命周期。时间分辨 X 射线吸收光谱显示,在反应条件下形成的钯簇对高 CO 氧化活性非常重要。结合密度泛函理论计算,我们得出结论:理想的钯簇大小约为 10-30 个钯原子。簇的形成和稳定性受应用温度和反应条件的影响。我们发现,在低于 200 °C 的温度下,贫反应进料中 1000 ppm CO 的短脉冲就能引发钯烧结,而在更高温度下,氧化过程占主导地位。环境透射电子显微镜揭示了在氧气环境中较高温度(400-500 °C)下的再分散过程,这导致了单个位点的形成,从而失去了活性。然而,由于一氧化碳的还原性,在反应气氛中冷却后又会形成团簇,从而结束了催化循环。利用已获得的有关钯簇生命周期的知识,我们系统地研究了催化剂组成对簇形成趋势的影响。通过 DRIFTS 测量发现,在反应条件下,Pd 与 CeO2 的比率似乎是 Pd 团聚的关键描述因子。虽然钯的负载量越高,形成团聚的概率就越大,但 CeO2 含量越高,就越容易形成氧化分散的钯。根据我们的研究结果,CeO2-Al2O3 支承催化剂的 Pd:CeO2 重量比为 1:10,在贫液条件下具有最高的一氧化碳氧化活性,与应用的合成方法无关。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

ACS Catalysis
CHEMISTRY, PHYSICAL-
CiteScore
20.80
自引率
6.20%
发文量
1253
审稿时长
1.5 months
期刊介绍:
ACS Catalysis is an esteemed journal that publishes original research in the fields of heterogeneous catalysis, molecular catalysis, and biocatalysis. It offers broad coverage across diverse areas such as life sciences, organometallics and synthesis, photochemistry and electrochemistry, drug discovery and synthesis, materials science, environmental protection, polymer discovery and synthesis, and energy and fuels.
The scope of the journal is to showcase innovative work in various aspects of catalysis. This includes new reactions and novel synthetic approaches utilizing known catalysts, the discovery or modification of new catalysts, elucidation of catalytic mechanisms through cutting-edge investigations, practical enhancements of existing processes, as well as conceptual advances in the field. Contributions to ACS Catalysis can encompass both experimental and theoretical research focused on catalytic molecules, macromolecules, and materials that exhibit catalytic turnover.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


