Total Synthesis of Chalaniline A: An Aminofulvene Fused Chromone from Vorinostat-Treated Fungus Chalara sp. 6661.
IF 3.3
2区 化学
Q1 CHEMISTRY, ORGANIC
引用次数: 0
Abstract
Chalaniline A, an aminofulveno[1,2-b]chromone derivative previously isolated from a vorinostat-treated ascomycete Chalara sp., was prepared in nine steps from orcinol (3,5-dihydroxytoluene). In a key transformation, the tricyclic ring system of the target was generated by a pyrrolidine-catalyzed double annulation between α-(methylsulfinyl)-2,6-dihydroxy-4-methylacetophenone and the ketaldoester, methyl 2,5-dioxopentanoate. The resulting tertiary alcohol (coniochaetone H) was further converted to chalaniline A by operations including dehydration (to yield a hydroxyfulvene), Vilsmeier reaction, and enamine exchange.
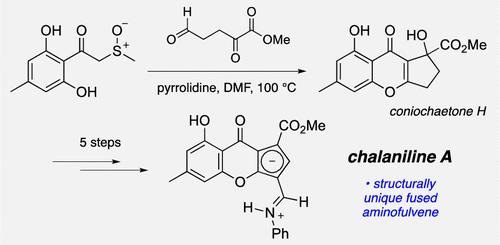
Chalaniline A 的全合成:来自 Vorinostat 处理过的真菌 Chalara sp.
Chalaniline A 是一种氨基富烯并[1,2-b]铬酮衍生物,以前从经伏立诺他处理的伞菌 Chalara sp.中分离出来,由 orcinol(3,5-二羟基甲苯)经九个步骤制备而成。在一个关键的转化过程中,目标物的三环系统是通过吡咯烷催化的 α-(甲亚磺酰基)-2,6-二羟基-4-甲基苯乙酮与酮酯 2,5- 二氧代戊酸甲酯之间的双环化反应生成的。通过脱水(生成羟基富烯)、维尔斯梅尔反应和烯胺交换等操作,将生成的叔醇(海茴香酮 H)进一步转化为查拉尼林 A。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

The Journal of Organic Chemistry
化学-有机化学
CiteScore
6.20
自引率
11.10%
发文量
1467
审稿时长
2 months
期刊介绍:
The Journal of Organic Chemistry welcomes original contributions of fundamental research in all branches of the theory and practice of organic chemistry. In selecting manuscripts for publication, the editors place emphasis on the quality and novelty of the work, as well as the breadth of interest to the organic chemistry community.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


