Proteogenomic characterization identifies clinical subgroups in EGFR and ALK wild-type never-smoker lung adenocarcinoma
IF 9.5
2区 医学
Q1 BIOCHEMISTRY & MOLECULAR BIOLOGY
引用次数: 0
Abstract
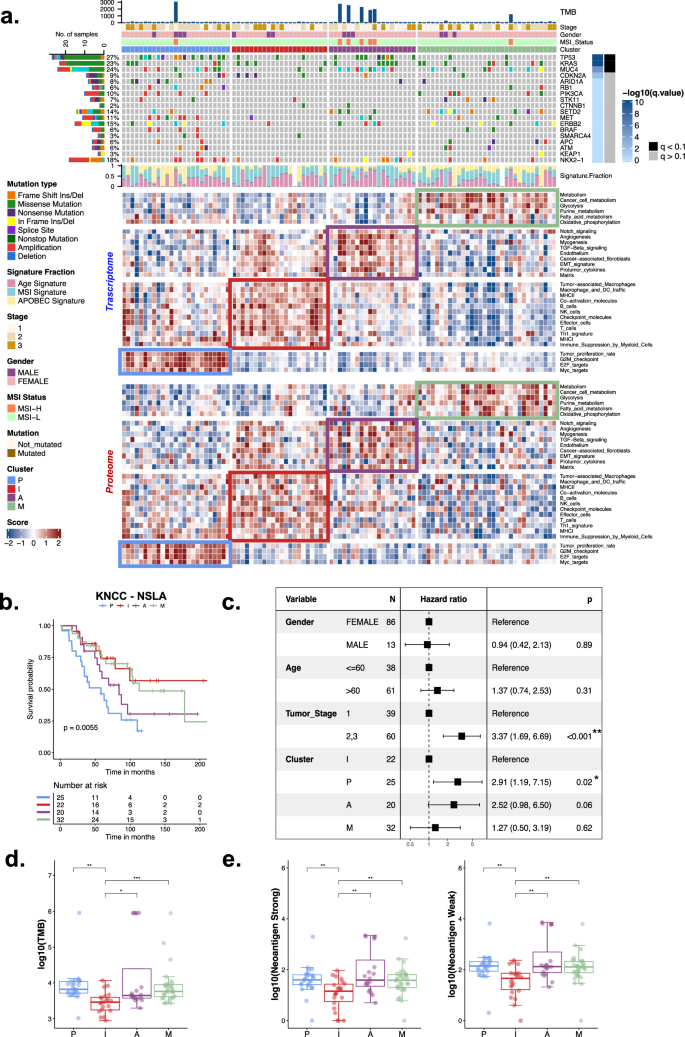
Patients with lung adenocarcinoma who have never smoked (NSLA) and lack key driver mutations, such as those in the EGFR and ALK genes, face limited options for targeted therapies. They also tend to have poorer outcomes with immune checkpoint inhibitors than lung cancer patients who have a history of smoking. The proteogenomic profile of nonsmoking lung adenocarcinoma patients without these oncogenic driver mutations is poorly understood, which complicates the precise molecular classification of these cancers and highlights a significant area of unmet clinical need. This study analyzed the genome, transcriptome, and LC‒MS/MS-TMT-driven proteome data of tumors obtained from 99 Korean never-smoker lung adenocarcinoma patients. NSLA tumors without EGFR or ALK driver oncogenes were classified into four proteogenomic subgroups: proliferation, angiogenesis, immune, and metabolism subgroups. These 4 molecular subgroups were strongly associated with distinct clinical outcomes. The proliferation and angiogenesis subtypes were associated with a poorer prognosis, while the immune subtype was associated with the most favorable outcome, which was validated in an external lung cancer dataset. Genomic-wide impacts were analyzed, and significant correlations were found between copy number alterations and both the transcriptome and proteome for several genes, with enrichment in the ERBB, neurotrophin, insulin, and MAPK signaling pathways. Proteogenomic analyses suggested several targetable genes and proteins, including CDKs and ATR, as potential therapeutic targets in the proliferation subgroup. Upregulated cytokines, such as CCL5 and CXCL13, in the immune subgroup may serve as potential targets for combination immunotherapy. Our comprehensive proteogenomic analysis revealed the molecular subtypes of EGFR- and ALK-wild-type NSLA with significant unmet clinical needs. Lung cancer is the leading cause of cancer deaths worldwide, with increasing cases in non-smokers, particularly Asian women. This research investigates lung adenocarcinoma in non-smokers who don’t have common genetic changes, using a multi-omics approach. The study involved 99 patients, specifically those without typical EGFR or ALK mutations, to better understand the disease at a molecular level and find new treatments. The study shows the variety within non-smoker lung cancers and suggests that different groups may need specific treatments. Understanding the molecular types of lung adenocarcinoma in non-smokers can lead to better, personalized treatments and improved health outcomes. This research could lead to more effective treatments for non-smoker lung cancer, potentially improving survival and quality of life for this growing patient group. This summary was initially drafted using artificial intelligence, then revised and fact-checked by the author.
蛋白质基因组特征识别表皮生长因子受体(EGFR)和 ALK 野生型从不吸烟者肺腺癌的临床亚组
从未吸烟(NSLA)且缺乏表皮生长因子受体(EGFR)和ALK基因等关键驱动基因突变的肺腺癌患者面临着靶向治疗的有限选择。与有吸烟史的肺癌患者相比,他们使用免疫检查点抑制剂的疗效也往往较差。人们对没有这些致癌驱动基因突变的非吸烟肺腺癌患者的蛋白质基因组概况知之甚少,这使得这些癌症的精确分子分类变得复杂,并凸显了一个尚未满足临床需求的重要领域。本研究分析了 99 名韩国从不吸烟肺腺癌患者肿瘤的基因组、转录组和 LC-MS/MS-TMT 驱动的蛋白质组数据。没有表皮生长因子受体(EGFR)或ALK驱动癌基因的NSLA肿瘤被分为4个蛋白质组亚组:增殖亚组、血管生成亚组、免疫亚组和代谢亚组。这4个分子亚组与不同的临床结果密切相关。增殖亚型和血管生成亚型与较差的预后相关,而免疫亚型与最有利的预后相关,这一点在外部肺癌数据集中得到了验证。对全基因组影响进行了分析,发现拷贝数改变与多个基因的转录组和蛋白质组之间存在显著相关性,ERBB、神经营养素、胰岛素和MAPK信号通路中的相关基因更为丰富。蛋白质基因组分析表明,包括CDK和ATR在内的几个可靶向基因和蛋白质是增殖亚组的潜在治疗靶点。免疫亚组中上调的细胞因子,如CCL5和CXCL13,可作为联合免疫疗法的潜在靶点。我们的综合蛋白基因组分析揭示了表皮生长因子受体(EGFR)型和ALK-wild型NSLA的分子亚型,这些亚型的重大临床需求尚未得到满足。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

Experimental and Molecular Medicine
医学-生化与分子生物学
CiteScore
19.50
自引率
0.80%
发文量
166
审稿时长
3 months
期刊介绍:
Experimental & Molecular Medicine (EMM) stands as Korea's pioneering biochemistry journal, established in 1964 and rejuvenated in 1996 as an Open Access, fully peer-reviewed international journal. Dedicated to advancing translational research and showcasing recent breakthroughs in the biomedical realm, EMM invites submissions encompassing genetic, molecular, and cellular studies of human physiology and diseases. Emphasizing the correlation between experimental and translational research and enhanced clinical benefits, the journal actively encourages contributions employing specific molecular tools. Welcoming studies that bridge basic discoveries with clinical relevance, alongside articles demonstrating clear in vivo significance and novelty, Experimental & Molecular Medicine proudly serves as an open-access, online-only repository of cutting-edge medical research.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


