Physical Properties of Renewable Solvents Cyrene, Dimethylisosorbide, γ-Valerolactone, Cyclopentylmethyl Ether, and 2-Methyltetrahydrofuran
IF 2
3区 工程技术
Q3 CHEMISTRY, MULTIDISCIPLINARY
引用次数: 0
Abstract
Renewable solvents derived from biomass have been regarded as promising alternatives to conventional organic solvents. A knowledge of the physical properties is crucial to accelerate the potential applications of a solvent, especially in industrial setups. Herein, we report water miscibility, surface tension (γ), density (ρ), dynamic viscosity (η), and refractive index (nD) of five structurally different renewable solvents: cyrene, dimethylisosorbide (DMI), γ-valerolactone (GVL), cyclopentylmethyl ether (CPME), and 2-methyltetrahydrofuran (2-MeTHF). Density and dynamic viscosity are measured in the temperature range of 283.15–363.15 K and surface tension is measured in the range of 298.15–363.15 K. Water miscibility and refractive index are estimated under ambient conditions. Comparatively higher water miscibility of cyrene, DMI, and GVL hints toward the availability of a greater number of H-bonding sites. At 298.15 K, all physical properties frame similar trends, with cyrene being at the top followed by DMI and GVL with CPME and 2-MeTHF at the bottom. Entropy of surface formation (Sγ) and enthalpy of surface formation (Hγ) were estimated using temperature dependence of surface tension. Dynamic viscosity follows Arrhenius-like expression with the temperature. The role of structural organization and the strength of intermolecular forces of interactions in controlling physical properties of renewable solvents is established.
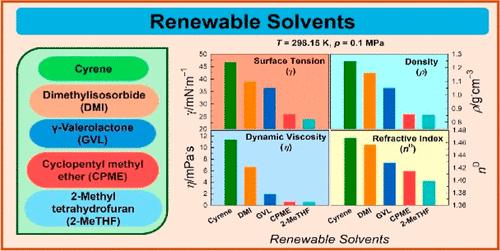
可再生溶剂芘、二甲基异山梨醇苷、γ-戊内酯、环戊基甲基醚和 2-甲基四氢呋喃的物理性质
从生物质中提取的可再生溶剂一直被视为传统有机溶剂的理想替代品。对物理特性的了解对于加快溶剂的潜在应用至关重要,尤其是在工业装置中。在此,我们报告了五种结构不同的可再生溶剂:芘、二甲基异山梨醇醚(DMI)、γ-戊内酯(GVL)、环戊基甲基醚(CPME)和 2-甲基四氢呋喃(2-MeTHF)的水混溶性、表面张力(γ)、密度(ρ)、动态粘度(η)和折射率(nD)。在 283.15-363.15 K 的温度范围内测量了密度和动态粘度,在 298.15-363.15 K 的温度范围内测量了表面张力。芘、DMI 和 GVL 的水混溶性相对较高,这表明存在更多的 H 键位点。在 298.15 K 时,所有物理性质都呈现出类似的趋势,芘的物理性质最高,其次是 DMI 和 GVL,CPME 和 2-MeTHF 的物理性质最低。利用表面张力的温度依赖性估算了表面形成熵(Sγ)和表面形成焓(Hγ)。动态粘度随温度的变化呈类似阿伦尼乌斯的表达式。确定了结构组织和分子间相互作用力的强度在控制可再生溶剂物理性质方面的作用。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

Journal of Chemical & Engineering Data
工程技术-工程:化工
CiteScore
5.20
自引率
19.20%
发文量
324
审稿时长
2.2 months
期刊介绍:
The Journal of Chemical & Engineering Data is a monthly journal devoted to the publication of data obtained from both experiment and computation, which are viewed as complementary. It is the only American Chemical Society journal primarily concerned with articles containing data on the phase behavior and the physical, thermodynamic, and transport properties of well-defined materials, including complex mixtures of known compositions. While environmental and biological samples are of interest, their compositions must be known and reproducible. As a result, adsorption on natural product materials does not generally fit within the scope of Journal of Chemical & Engineering Data.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


