Regulating H2S release from self-assembled peptide H2S-donor conjugates using cysteine derivatives†
IF 2.9
3区 化学
Q1 CHEMISTRY, ORGANIC
引用次数: 0
Abstract
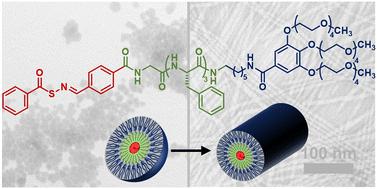
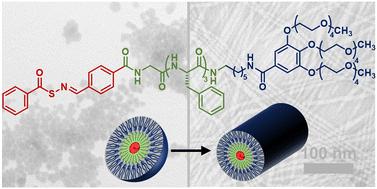
Self-assembled peptides provide a modular and diverse platform for drug delivery, and innovative delivery methods are needed for delivery of hydrogen sulfide (H2S), an endogenous signaling molecule (gasotransmitter) with significant therapeutic potential. Of the available types of H2S donors, peptide/protein H2S donor conjugates (PHDCs) offer significant versatility. Here we discuss the design, synthesis, and in-depth study of a PHDC containing three covalently linked components: a thiol-triggered H2S donor based on an S-aroylthiooxime (SATO), a GFFF tetrapeptide, and a tetraethylene glycol (TEG) dendron. Conventional transmission electron microscopy showed that the PHDC self-assembled into spherical structures without heat or stirring, but it formed nanofibers with gentle heat (37 °C) and stirring. Circular dichroism (CD) spectroscopy data collected during self-assembly under nanofiber-forming conditions suggested an increase in β-sheet character and a decrease in organization of the SATO units. Release of H2S from the nanofibers was studied through triggering with various thiols. The release rate and total amount of H2S released over both short (5 h) and long (7 d) time scales varied with the charge state: negatively charged and zwitterionic thiols (e.g., Ac-Cys-OH and H-Cys-OH) triggered release slowly while a neutral thiol (Ac-Cys-OMe) showed ∼10-fold faster release, and a positively charged thiol (H-Cys-OMe) triggered H2S release nearly 50-fold faster than the negatively charged thiols. CD spectroscopy studies monitoring changes in secondary structure over time during H2S release showed similar trends. This study sheds light on the driving forces behind self-assembling nanostructures and offers insights into tuning H2S release through thiol charge state modulation.
利用半胱氨酸衍生物调节自组装肽 H2S-供体共轭物中 H2S 的释放
自组装肽为药物输送提供了一个模块化和多样化的平台,而硫化氢(H2S)是一种具有巨大治疗潜力的内源性信号分子(气体递质),因此需要创新的输送方法来输送硫化氢。在现有的 H2S 给体类型中,肽/蛋白质 H2S 给体共轭物(PHDCs)具有显著的多功能性。在此,我们讨论了一种 PHDC 的设计、合成和深入研究,该 PHDC 包含三种共价连接的成分:基于 S-aroylthiooxime (SATO)的硫醇触发 H2S 给体、GFFF 四肽和四甘醇(TEG)树枝状化合物。传统的透射电子显微镜显示,PHDC 无需加热或搅拌就能自组装成球形结构,但在轻微加热(37 °C)和搅拌的情况下就能形成纳米纤维。在纳米纤维形成条件下自组装过程中收集的圆二色性(CD)光谱数据表明,β-片状特征增加,SATO 单元的组织减少。通过使用各种硫醇进行触发,研究了纳米纤维释放 H2S 的情况。在短(5 小时)和长(7 天)时间范围内,H2S 的释放速度和释放总量随电荷状态而变化:带负电荷和齐聚离子硫醇(如 Ac-Cys-OH 和 H-Cys-OH)触发释放的速度较慢,而中性硫醇(Ac-Cys-OMe)触发释放的速度快 10 倍;带正电荷的硫醇(H-Cys-OMe)触发释放 H2S 的速度比带负电荷的硫醇快近 50 倍。监测 H2S 释放过程中二级结构随时间变化的 CD 光谱研究也显示了类似的趋势。这项研究揭示了自组装纳米结构背后的驱动力,并为通过调节硫醇电荷状态来调整 H2S 释放提供了启示。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

Organic & Biomolecular Chemistry
化学-有机化学
CiteScore
5.50
自引率
9.40%
发文量
1056
审稿时长
1.3 months
期刊介绍:
Organic & Biomolecular Chemistry is an international journal using integrated research in chemistry-organic chemistry. Founded in 2003 by the Royal Society of Chemistry, the journal is published in Semimonthly issues and has been indexed by SCIE, a leading international database. The journal focuses on the key research and cutting-edge progress in the field of chemistry-organic chemistry, publishes and reports the research results in this field in a timely manner, and is committed to becoming a window and platform for rapid academic exchanges among peers in this field. The journal's impact factor in 2023 is 2.9, and its CiteScore is 5.5.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


