Streamlined screening platforms lead to the discovery of pachysiphine synthase from Tabernanthe iboga
IF 8.3
1区 生物学
Q1 PLANT SCIENCES
引用次数: 0
Abstract
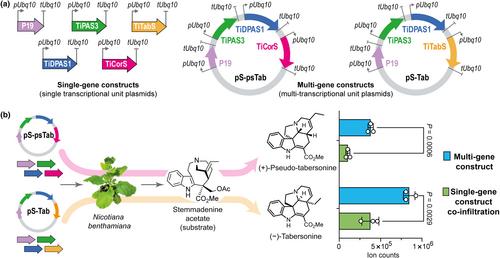
通过简化筛选平台,从伊博加(Tabernanthe iboga)中发现茯苓碱合成酶
摘要 植物专一性代谢主要由细胞色素 P450(CYP450s)酶催化的关键化学支架的氧化定制驱动。在药用植物 Tabernanthe iboga(俗称伊博格)中发现的单萜吲哚生物碱(MIAs)tabersonine 和 pseudo-tabersonine,是通过氧化作用定制的,所涉及的酶仍然未知。在此,我们开发了一种简化的筛选策略,以测试烟草中 T. iboga CYP450s 的活性。利用多基因构建体编码塔巴索宁和假塔巴索宁支架的生物合成,我们旨在发现负责这些支架氧化转化的 CYP450s。我们的方法确定了两种 T. iboga 细胞色素 P450 酶:茯苓碱合成酶(PS)和 16-hydroxy-tabersonine 合成酶(T16H)。这些酶催化塔巴索碱的环氧化反应和特定位点羟基化反应,分别生成茯苓碱和 16-OH-塔巴索碱。这项工作为了解 MIAs 的生物合成途径提供了新的视角,并强调了将 N. benthamiana 和 Catharanthus roseus 作为植物酶功能表征平台的实用性。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

New Phytologist
生物-植物科学
自引率
5.30%
发文量
728
期刊介绍:
New Phytologist is an international electronic journal published 24 times a year. It is owned by the New Phytologist Foundation, a non-profit-making charitable organization dedicated to promoting plant science. The journal publishes excellent, novel, rigorous, and timely research and scholarship in plant science and its applications. The articles cover topics in five sections: Physiology & Development, Environment, Interaction, Evolution, and Transformative Plant Biotechnology. These sections encompass intracellular processes, global environmental change, and encourage cross-disciplinary approaches. The journal recognizes the use of techniques from molecular and cell biology, functional genomics, modeling, and system-based approaches in plant science. Abstracting and Indexing Information for New Phytologist includes Academic Search, AgBiotech News & Information, Agroforestry Abstracts, Biochemistry & Biophysics Citation Index, Botanical Pesticides, CAB Abstracts®, Environment Index, Global Health, and Plant Breeding Abstracts, and others.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


