Antagonism between H3K27me3 and genome–lamina association drives atypical spatial genome organization in the totipotent embryo
IF 31.7
1区 生物学
Q1 GENETICS & HEREDITY
引用次数: 0
Abstract
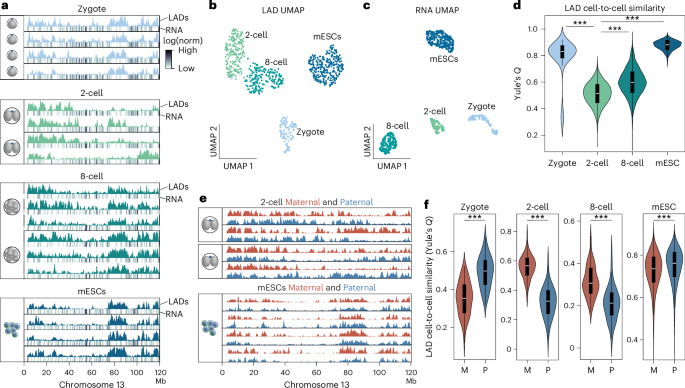
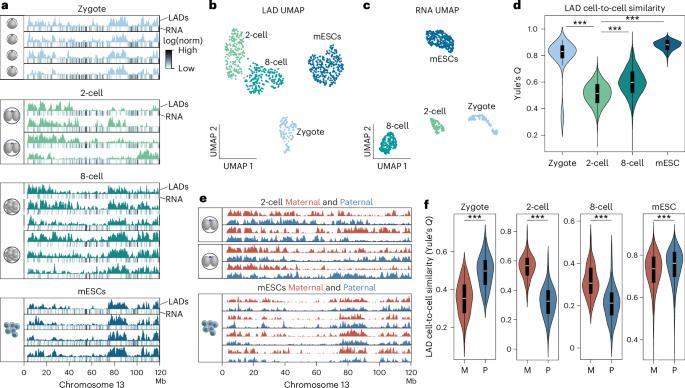
In mammals, early embryonic development exhibits highly unusual spatial positioning of genomic regions at the nuclear lamina, but the mechanisms underpinning this atypical genome organization remain elusive. Here, we generated single-cell profiles of lamina-associated domains (LADs) coupled with transcriptomics, which revealed a striking overlap between preimplantation-specific LAD dissociation and noncanonical broad domains of H3K27me3. Loss of H3K27me3 resulted in a restoration of canonical LAD profiles, suggesting an antagonistic relationship between lamina association and H3K27me3. Tethering of H3K27me3 to the nuclear periphery showed that the resultant relocalization is partially dependent on the underlying DNA sequence. Collectively, our results suggest that the atypical organization of LADs in early developmental stages is the result of a tug-of-war between intrinsic affinity for the nuclear lamina and H3K27me3, constrained by the available space at the nuclear periphery. This study provides detailed insight into the molecular mechanisms regulating nuclear organization during early mammalian development. Single-cell profiling of lamina-associated domains (LADs) during early mouse development reveals an overlap between preimplantation-specific LAD dissociation and noncanonical broad H3K27me3 domains. Loss of H3K27me3 restores canonical LAD profiles.
H3K27me3 与基因组-拉米娜关联之间的拮抗作用驱动全能胚胎的非典型空间基因组组织
在哺乳动物中,早期胚胎发育表现出基因组区域在核薄层极不寻常的空间定位,但这种非典型基因组组织的基础机制仍然难以捉摸。在这里,我们结合转录组学生成了薄层相关结构域(LADs)的单细胞图谱,发现胚胎植入前特异性LAD解离与H3K27me3的非典型宽域之间存在惊人的重叠。缺失H3K27me3会导致典型LAD图谱的恢复,这表明薄片关联与H3K27me3之间存在拮抗关系。将 H3K27me3 拴系到核外围表明,由此产生的重新定位部分依赖于底层 DNA 序列。总之,我们的研究结果表明,LADs 在早期发育阶段的非典型组织是核薄层固有亲和力与 H3K27me3 之间角力的结果,受到核外围可用空间的限制。这项研究详细揭示了哺乳动物早期发育过程中调节核组织的分子机制。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

Nature genetics
生物-遗传学
CiteScore
43.00
自引率
2.60%
发文量
241
审稿时长
3 months
期刊介绍:
Nature Genetics publishes the very highest quality research in genetics. It encompasses genetic and functional genomic studies on human and plant traits and on other model organisms. Current emphasis is on the genetic basis for common and complex diseases and on the functional mechanism, architecture and evolution of gene networks, studied by experimental perturbation.
Integrative genetic topics comprise, but are not limited to:
-Genes in the pathology of human disease
-Molecular analysis of simple and complex genetic traits
-Cancer genetics
-Agricultural genomics
-Developmental genetics
-Regulatory variation in gene expression
-Strategies and technologies for extracting function from genomic data
-Pharmacological genomics
-Genome evolution
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


