Enhanced Nitric Oxide Delivery Through Self‐Assembling Nanoparticles for Eradicating Gram‐Negative Bacteria
IF 10
2区 医学
Q1 ENGINEERING, BIOMEDICAL
引用次数: 0
Abstract
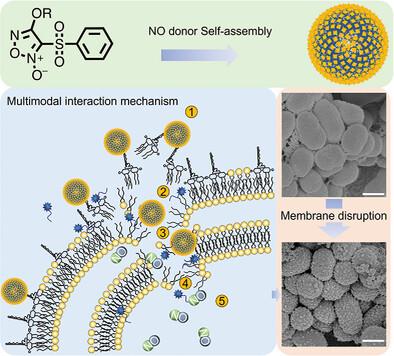
In the current battle against antibiotic resistance, the resilience of Gram‐negative bacteria against traditional antibiotics is due not only to their protective outer membranes but also to mechanisms like efflux pumps and enzymatic degradation of drugs, underscores the urgent need for innovative antimicrobial tactics. Herein, this study presents an innovative method involving the synthesis of three furoxan derivatives engineered to self‐assemble into nitric oxide (NO) donor nanoparticles (FuNPs). These FuNPs, notably supplied together with polymyxin B (PMB), achieve markedly enhanced bactericidal efficacy against a wide spectrum of bacterial phenotypes at considerably lower NO concentrations (0.1–2.8 µg mL
通过自组装纳米粒子增强一氧化氮输送以消灭革兰氏阴性菌
在当前与抗生素耐药性的斗争中,革兰氏阴性细菌对传统抗生素的抵抗力不仅取决于其保护性外膜,还取决于药物的外排泵和酶降解等机制,因此迫切需要创新的抗菌策略。在此,本研究提出了一种创新方法,涉及合成三种呋喃类衍生物,并将其设计为能自我组装成一氧化氮(NO)供体纳米粒子(FuNPs)。这些 FuNPs 特别是与多粘菌素 B (PMB) 一起使用时,在相当低的一氧化氮浓度(0.1-2.8 µg mL-1)下对多种细菌表型的杀菌效力明显增强,这比一氧化氮供体的报告数据(≥200 µg mL-1)至少低十倍。利用共焦、扫描和透射电子显微镜技术阐明了杀菌机制。中子反射仪证实,FuNPs 通过与细菌表面的多糖特异性结合,导致结构紊乱,从而引发膜破坏。随后,PMB 与外膜上的脂质 A 结合,增强了渗透性,从而与 FuNPs 形成协同杀菌作用。这一开创性战略强调了自组装在 NO 递送中的实用性,是规避传统抗生素耐药性障碍的开创性范例,标志着下一代抗菌剂开发的重大飞跃。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

Advanced Healthcare Materials
工程技术-生物材料
CiteScore
14.40
自引率
3.00%
发文量
600
审稿时长
1.8 months
期刊介绍:
Advanced Healthcare Materials, a distinguished member of the esteemed Advanced portfolio, has been dedicated to disseminating cutting-edge research on materials, devices, and technologies for enhancing human well-being for over ten years. As a comprehensive journal, it encompasses a wide range of disciplines such as biomaterials, biointerfaces, nanomedicine and nanotechnology, tissue engineering, and regenerative medicine.
文献相关原料
| 公司名称 | 产品信息 | 采购帮参考价格 |
|---|
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


