Manganese(II) oxide-embedded dopamine-derived carbon nanospheres for durable zinc-ion batteries
IF 6
2区 材料科学
Q1 CHEMISTRY, MULTIDISCIPLINARY
引用次数: 0
Abstract
Manganese oxides are considered highly promising as cathode materials for aqueous zinc-ion batteries (ZIBs) owing to their abundant resources, high discharge potential, and substantial theoretical capacity. Nonetheless, MnO is commonly perceived to exhibit insufficient electrochemical activity and is deemed unsuitable for Zn2+ storage. Herein, MnO-embedded PDA-derived carbon (MnO/C-PDA) is utilized as the cathode material for ZIBs, and its electrochemical behavior in ZnSO4 electrolytes with varying MnSO4 concentrations is investigated. The results indicate that the incorporation of manganese salt electrolyte notably enhances electrode capacity, though excessively high concentrations of manganese salt diminish electrode activity. In the electrolyte containing 0.2 M MnSO4, MnO-C/PDA exhibits a capacity of 295.4 mA h g−1 at 0.1 A g−1, with negligible capacity degradation even after 100 cycles. Ex situ characterization reveals that during the charging process, MnO transformed into amorphous MnOx, accompanied by the deposition of manganese salts forming MnOx, while the discharge process involved the co-insertion of Zn2+ and H+. This work is anticipated to enhance comprehension regarding the charge and discharge mechanisms of MnO, thus aiding in the development of manganese oxide cathodes tailored for ZIBs.
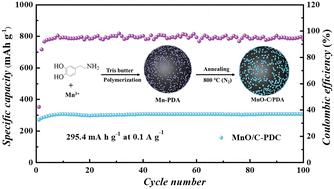
用于耐用锌-离子电池的嵌入式多巴胺衍生碳纳米氧化锰(II)球
锰氧化物资源丰富、放电电位高、理论容量大,因此被认为非常有希望成为水性锌离子电池(ZIB)的阴极材料。然而,人们普遍认为氧化锰的电化学活性不足,不适合储存 Zn2+。本文利用 MnO 嵌入 PDA 衍生碳(MnO/C-PDA)作为 ZIB 的阴极材料,并研究了其在不同 MnSO4 浓度的 ZnSO4 电解质中的电化学行为。结果表明,加入锰盐电解质可显著提高电极容量,但锰盐浓度过高会降低电极活性。在含有 0.2 M MnSO4 的电解液中,MnO-C/PDA 在 0.1 A g-1 的条件下显示出 295.4 mA h g-1 的容量,即使循环 100 次后,容量衰减也可以忽略不计。原位表征显示,在充电过程中,MnO 转变为无定形的 MnOx,同时锰盐沉积形成 MnOx,而放电过程则涉及 Zn2+ 和 H+ 的共同插入。这项研究有望加深人们对氧化锰充放电机理的理解,从而帮助开发适合 ZIB 的氧化锰阴极。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

Materials Chemistry Frontiers
Materials Science-Materials Chemistry
CiteScore
12.00
自引率
2.90%
发文量
313
期刊介绍:
Materials Chemistry Frontiers focuses on the synthesis and chemistry of exciting new materials, and the development of improved fabrication techniques. Characterisation and fundamental studies that are of broad appeal are also welcome.
This is the ideal home for studies of a significant nature that further the development of organic, inorganic, composite and nano-materials.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


