Development of a First-in-Class DNMT1/HDAC Inhibitor with Improved Therapeutic Potential and Potentiated Antitumor Immunity
IF 6.8
1区 医学
Q1 CHEMISTRY, MEDICINAL
引用次数: 0
Abstract
Epigenetic therapies have emerged as a key paradigm for treating malignancies. In this study, a series of DNMT1/HDAC dual inhibitors were obtained by fusing the key pharmacophores from DNMT1 inhibitors (DNMT1i) and HDAC inhibitors (HDACi). Among them, compound (R)-23a demonstrated significant DNMT1 and HDAC inhibition both in vitro and in cells and largely phenocopied the synergistic effects of combined DNMT1i and HDACi in reactivating epigenetically silenced tumor suppressor genes (TSGs). This translated into a profound tumor growth inhibition (TGI = 98%) of (R)-23a in an MV-4-11 xenograft model, while displaying improved tolerability compared with single agent combination. Moreover, in a syngeneic MC38 mouse colorectal tumor model, (R)-23a outperformed the combinatory treatment in reshaping the tumor immune microenvironment and inducing tumor regression. Collectively, the novel DNMT1/HDAC dual inhibitor (R)-23a effectively reverses the cancer-specific epigenetic abnormalities and holds great potential for further development into cancer therapeutic agents.
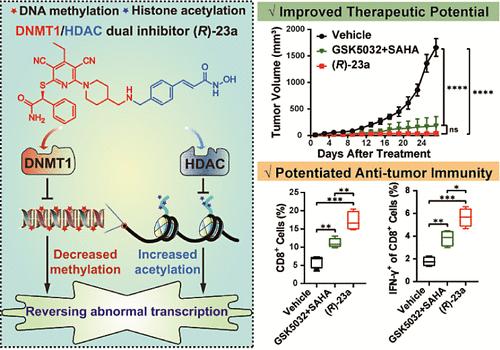
开发出治疗潜力更大、抗肿瘤免疫力更强的一流 DNMT1/HDAC 抑制剂
表观遗传疗法已成为治疗恶性肿瘤的关键范例。本研究通过融合 DNMT1 抑制剂(DNMT1i)和 HDAC 抑制剂(HDACi)的关键药效团,获得了一系列 DNMT1/HDAC 双抑制剂。其中,化合物(R)-23a 在体外和细胞内均表现出显著的 DNMT1 和 HDAC 抑制作用,并在很大程度上表征了 DNMT1i 和 HDACi 联合抑制剂在重新激活表观遗传沉默的肿瘤抑制基因(TSGs)方面的协同作用。在 MV-4-11 异种移植模型中,(R)-23a 可显著抑制肿瘤生长(TGI = 98%),同时与单药组合相比,耐受性更好。此外,在合成MC38小鼠结直肠肿瘤模型中,(R)-23a在重塑肿瘤免疫微环境和诱导肿瘤消退方面的表现优于联合疗法。总之,新型 DNMT1/HDAC 双抑制剂 (R)-23a 能有效逆转癌症特异性表观遗传异常,具有进一步开发为癌症治疗药物的巨大潜力。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

Journal of Medicinal Chemistry
医学-医药化学
CiteScore
4.00
自引率
11.00%
发文量
804
审稿时长
1.9 months
期刊介绍:
The Journal of Medicinal Chemistry is a prestigious biweekly peer-reviewed publication that focuses on the multifaceted field of medicinal chemistry. Since its inception in 1959 as the Journal of Medicinal and Pharmaceutical Chemistry, it has evolved to become a cornerstone in the dissemination of research findings related to the design, synthesis, and development of therapeutic agents.
The Journal of Medicinal Chemistry is recognized for its significant impact in the scientific community, as evidenced by its 2022 impact factor of 7.3. This metric reflects the journal's influence and the importance of its content in shaping the future of drug discovery and development. The journal serves as a vital resource for chemists, pharmacologists, and other researchers interested in the molecular mechanisms of drug action and the optimization of therapeutic compounds.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


