Production of Distinct Fibrillar, Oligomeric, and Other Aggregation States from Network Models of Multibody Interaction
IF 5.7
1区 化学
Q2 CHEMISTRY, PHYSICAL
引用次数: 0
Abstract
Protein aggregation can produce a wide range of states, ranging from fibrillar structures and oligomers to unstructured and semistructured gel phases. Recent work has shown that many of these states can be recapitulated by relatively simple, topological models specified in terms of multibody interaction energies, providing a direct connection between aggregate intermolecular forces and aggregation products. Here, we examine a low-dimensional network Hamiltonian model (NHM) based on four basic multibody interactions found in any aggregate system. We characterize the phase behavior of this NHM family, showing that fibrils arise from a balance between elongation-inducing and contact-inhibiting forces. Complex oligomers (including annular oligomers resembling those thought to be toxic species in Alzheimer’s disease) also form distinct phases in this regime, controlled in part by closure-inducing forces. We show that phase structure is largely independent of system size, and provide evidence of a rich structure of minor oligomeric phases that can arise from appropriate conditions. We characterize the phase behavior of this NHM family, demonstrating the range of ordered and disordered aggregation states possible with this set of interactions. As we show, fibrils arise from a balance between elongation-inducing and contact-inhibiting forces, existing in a regime bounded by gel-like and disaggregated phases; complex oligomers (including annular oligomers resembling those thought to be toxic species in Alzheimer’s disease) also form distinct phases in this regime, controlled in part by closure-inducing forces. We show that phase structure is largely independent of system size, allowing generalization to macroscopic systems, and provide evidence of a rich structure of minor oligomeric phases that can arise from appropriate conditions.
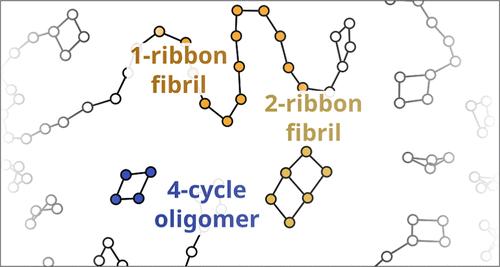
从多体相互作用网络模型中生成不同的纤维状、寡聚状和其他聚合状态
蛋白质聚集可产生多种状态,从纤维状结构和低聚物到非结构和半结构凝胶相。最近的研究表明,这些状态中的许多状态都可以通过相对简单的拓扑模型来重现,这些模型是根据多体相互作用能来指定的,从而提供了聚集分子间作用力与聚集产物之间的直接联系。在此,我们研究了一种低维网络哈密顿模型(NHM),该模型基于任何聚合体系中都能发现的四种基本多体相互作用。我们描述了这一 NHM 族的相行为特征,表明纤维是由伸长诱导力和接触抑制力之间的平衡产生的。复杂的低聚物(包括环状低聚物,类似于那些被认为是阿尔茨海默氏症毒性物质的低聚物)也会在这种机制中形成独特的相,部分由闭合诱导力控制。我们的研究表明,相结构在很大程度上与系统大小无关,并提供了在适当条件下可产生丰富的次要低聚物相结构的证据。我们描述了这个 NHM 家族的相行为特征,展示了这组相互作用可能产生的有序和无序聚集状态的范围。正如我们所展示的那样,纤丝产生于伸长诱导力和接触抑制力之间的平衡,存在于以凝胶状相和分解相为边界的体系中;复杂的低聚物(包括类似于阿尔茨海默氏症毒性物种的环状低聚物)也会在此体系中形成不同的相,部分由闭合诱导力控制。我们的研究表明,相结构在很大程度上与系统大小无关,因此可以推广到宏观系统,并提供了在适当条件下可以产生丰富的次要低聚物相结构的证据。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

Journal of Chemical Theory and Computation
化学-物理:原子、分子和化学物理
CiteScore
9.90
自引率
16.40%
发文量
568
审稿时长
1 months
期刊介绍:
The Journal of Chemical Theory and Computation invites new and original contributions with the understanding that, if accepted, they will not be published elsewhere. Papers reporting new theories, methodology, and/or important applications in quantum electronic structure, molecular dynamics, and statistical mechanics are appropriate for submission to this Journal. Specific topics include advances in or applications of ab initio quantum mechanics, density functional theory, design and properties of new materials, surface science, Monte Carlo simulations, solvation models, QM/MM calculations, biomolecular structure prediction, and molecular dynamics in the broadest sense including gas-phase dynamics, ab initio dynamics, biomolecular dynamics, and protein folding. The Journal does not consider papers that are straightforward applications of known methods including DFT and molecular dynamics. The Journal favors submissions that include advances in theory or methodology with applications to compelling problems.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


