DNA microbeads for spatio-temporally controlled morphogen release within organoids
IF 38.1
1区 材料科学
Q1 MATERIALS SCIENCE, MULTIDISCIPLINARY
引用次数: 0
Abstract
Organoids are transformative in vitro model systems that mimic features of the corresponding tissue in vivo. However, across tissue types and species, organoids still often fail to reach full maturity and function because biochemical cues cannot be provided from within the organoid to guide their development. Here we introduce nanoengineered DNA microbeads with tissue mimetic tunable stiffness for implementing spatio-temporally controlled morphogen gradients inside of organoids at any point in their development. Using medaka retinal organoids and early embryos, we show that DNA microbeads can be integrated into embryos and organoids by microinjection and erased in a non-invasive manner with light. Coupling a recombinant surrogate Wnt to the DNA microbeads, we demonstrate the spatio-temporally controlled morphogen release from the microinjection site, which leads to morphogen gradients resulting in the formation of retinal pigmented epithelium while maintaining neuroretinal cell types. Thus, we bioengineered retinal organoids to more closely mirror the cell type diversity of in vivo retinae. Owing to the facile, one-pot fabrication process, the DNA microbead technology can be adapted to other organoid systems for improved tissue mimicry. Creating precise morphogen gradients for tissue engineering is challenging. Here the authors present mechanically tunable DNA hydrogel-based microbeads for light-controlled morphogen release in retinal organoids for better tissue mimicry.
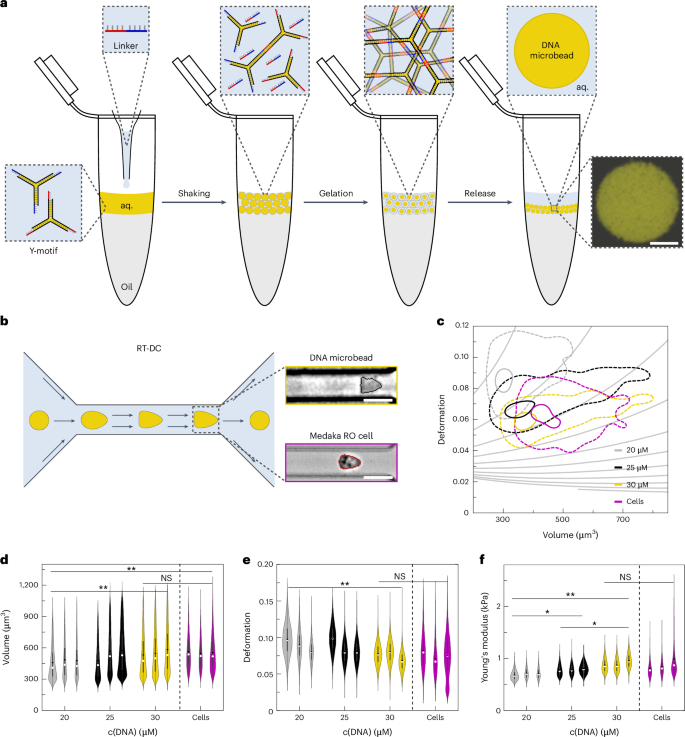

用于有机体内时空控制形态发生器释放的 DNA 微珠
类器官是一种可模拟体内相应组织特征的变革性体外模型系统。然而,在不同的组织类型和物种中,类器官仍常常无法完全成熟并发挥功能,因为类器官内部无法提供生化线索来指导其发育。在这里,我们引入了具有组织模拟可调硬度的纳米工程DNA微珠,用于在有机体发育的任何阶段在其内部实现时空可控的形态发生梯度。我们利用青鳉视网膜有机体和早期胚胎,证明了 DNA 微珠可以通过微注射集成到胚胎和有机体中,并用光照以非侵入方式清除。将重组替代 Wnt 与 DNA 微珠结合,我们证明了从微注射部位释放形态发生器的时空控制,这导致了形态发生器梯度,形成了视网膜色素上皮,同时保持了神经视网膜细胞类型。因此,我们对视网膜器官组织进行了生物工程改造,使其更接近于体内视网膜细胞类型的多样性。由于 DNA 微珠技术采用简便的单锅制造工艺,因此可应用于其他类器官系统,以提高组织仿真度。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

Nature nanotechnology
工程技术-材料科学:综合
CiteScore
59.70
自引率
0.80%
发文量
196
审稿时长
4-8 weeks
期刊介绍:
Nature Nanotechnology is a prestigious journal that publishes high-quality papers in various areas of nanoscience and nanotechnology. The journal focuses on the design, characterization, and production of structures, devices, and systems that manipulate and control materials at atomic, molecular, and macromolecular scales. It encompasses both bottom-up and top-down approaches, as well as their combinations.
Furthermore, Nature Nanotechnology fosters the exchange of ideas among researchers from diverse disciplines such as chemistry, physics, material science, biomedical research, engineering, and more. It promotes collaboration at the forefront of this multidisciplinary field. The journal covers a wide range of topics, from fundamental research in physics, chemistry, and biology, including computational work and simulations, to the development of innovative devices and technologies for various industrial sectors such as information technology, medicine, manufacturing, high-performance materials, energy, and environmental technologies. It includes coverage of organic, inorganic, and hybrid materials.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


