Distal activity patterns shape the spatial specificity of neurovascular coupling
IF 21.2
1区 医学
Q1 NEUROSCIENCES
引用次数: 0
Abstract
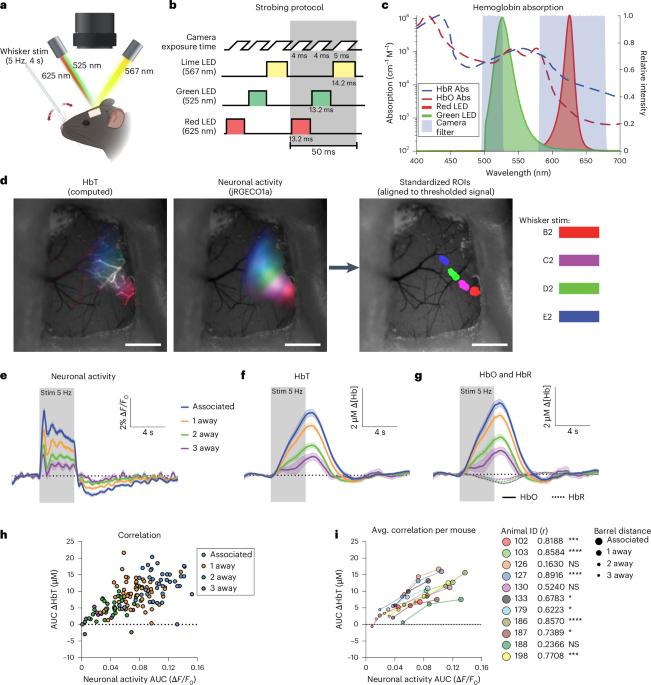
Neurovascular coupling links brain activity to local changes in blood flow, forming the basis for non-invasive brain mapping. Using multiscale imaging, we investigated how vascular activity spatially relates to neuronal activity elicited by single whiskers across different columns and layers of mouse cortex. Here we show that mesoscopic hemodynamic signals quantitatively reflect neuronal activity across space but are composed of a highly heterogeneous pattern of responses across individual vessel segments that is poorly predicted by local neuronal activity. Rather, this heterogeneity is dependent on vessel directionality, specifically in thalamocortical input layer 4, where capillaries respond preferentially to neuronal activity patterns along their downstream perfusion domain. Thus, capillaries fine-tune blood flow based on distant activity and encode laminar-specific activity patterns. These findings imply that vascular anatomy sets a resolution limit on functional imaging signals, where individual blood vessels inaccurately report neuronal activity in their immediate vicinity but, instead, integrate activity patterns along the vascular arbor. The spatial relationship between neuronal and vascular activity remains highly debated. In this study, the authors used multiscale optical imaging to show how vascular architecture limits the spatial specificity of neurovascular coupling.
远端活动模式决定了神经血管耦合的空间特异性
神经血管耦合将大脑活动与局部血流变化联系起来,为无创脑图绘制奠定了基础。利用多尺度成像技术,我们研究了血管活动与小鼠皮层不同列和层的单根胡须所引发的神经元活动之间的空间关系。我们在此表明,中观血液动力学信号定量地反映了整个空间的神经元活动,但这些信号是由单个血管节段的高度异质性反应模式组成的,而这种反应模式并不能很好地预测局部神经元活动。相反,这种异质性取决于血管的方向性,特别是在丘脑皮层输入第 4 层,毛细血管会优先响应沿其下游灌注域的神经元活动模式。因此,毛细血管根据远处的活动对血流进行微调,并编码特定层的活动模式。这些发现意味着,血管解剖学为功能成像信号设定了分辨率限制,单个血管不能准确报告其附近的神经元活动,而是要整合沿血管轴的活动模式。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

Nature neuroscience
医学-神经科学
CiteScore
38.60
自引率
1.20%
发文量
212
审稿时长
1 months
期刊介绍:
Nature Neuroscience, a multidisciplinary journal, publishes papers of the utmost quality and significance across all realms of neuroscience. The editors welcome contributions spanning molecular, cellular, systems, and cognitive neuroscience, along with psychophysics, computational modeling, and nervous system disorders. While no area is off-limits, studies offering fundamental insights into nervous system function receive priority.
The journal offers high visibility to both readers and authors, fostering interdisciplinary communication and accessibility to a broad audience. It maintains high standards of copy editing and production, rigorous peer review, rapid publication, and operates independently from academic societies and other vested interests.
In addition to primary research, Nature Neuroscience features news and views, reviews, editorials, commentaries, perspectives, book reviews, and correspondence, aiming to serve as the voice of the global neuroscience community.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


