Mixed-charge hyperbranched polymer nanoparticles with selective antibacterial action for fighting antimicrobial resistance
IF 9.4
1区 医学
Q1 ENGINEERING, BIOMEDICAL
引用次数: 0
Abstract
The escalating menace of antimicrobial resistance (AMR) presents a profound global threat to life and assets. However, the incapacity of metal ions/reactive oxygen species (ROS) or the indiscriminate intrinsic interaction of cationic groups to distinguish between bacteria and mammalian cells undermines the essential selectivity required in these nanomaterials for an ideal antimicrobial agent. Hence, we devised and synthesized a range of biocompatible mixed-charge hyperbranched polymer nanoparticles (MCHPNs) incorporating cationic, anionic, and neutral alkyl groups to effectively combat multidrug-resistant bacteria and mitigate AMR. This outcome stemmed from the structural, antibacterial activity, and biocompatibility analysis of seven MCHPNs, among which MCHPN7, with a ratio of cationic groups, anionic groups, and long alkyl chains at 27:59:14, emerged as the lead candidate. Importantly, owing to inherent differences in membrane potential among diverse species, alongside its nano-size (6–15 nm) and high hydrophilicity (Kow = 0.04), MCHPN7 exhibited exceptional selective bactericidal effects over mammalian cells (selectivity index > 564) in vitro and in vivo. By inducing physical membrane disruption, MCHPN7 effectively eradicated antibiotic-resistant bacteria and significantly delayed the emergence of bacterial resistance. Utilized as a coating, MCHPN7 endowed initially inert surfaces with the ability to impede biofilm formation and mitigate infection-related immune responses in mouse models. This research heralds the advent of biocompatible polymer nanoparticles and harbors significant implications in our ongoing combat against AMR.
Statement of significance
The escalating prevalence of antimicrobial resistance (AMR) has been acknowledged as one of the most significant threats to global health.
Therefore, a series of mixed-charge hyperbranched polymer nanoparticles (MCHPNs) with selective antibacterial action were designed and synthesized. Owing to inherent differences in membrane potential among diverse species and high hydrophilicity (Kow = 0.04), the optimal nanoparticles exhibited exceptional selective bactericidal effects over mammalian cells (selectivity index >564) and significantly delayed the emergence of bacterial resistance. Importantly, they endowed surfaces with the ability to impede biofilm formation and mitigate infection-related immune responses.
Furthermore, the above findings focus on addressing the problem of AMR in Post-Pandemic, which will for sure attract attention from both academic and industry research.
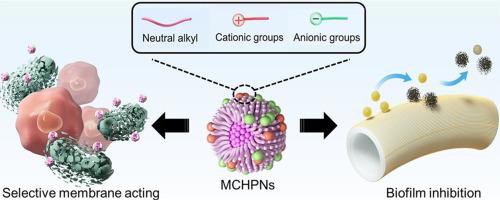
具有选择性抗菌作用的混合电荷超支化聚合物纳米粒子,用于对抗抗菌药耐药性。
抗菌药耐药性(AMR)的不断升级对全球生命和财产构成了严重威胁。然而,金属离子/活性氧(ROS)或阳离子基团不加区分的内在相互作用无法区分细菌和哺乳动物细胞,这就削弱了这些纳米材料作为理想抗菌剂所需的基本选择性。因此,我们设计并合成了一系列具有生物相容性的混合电荷超支化聚合物纳米粒子(MCHPNs),其中包含阳离子、阴离子和中性烷基,可有效抗击多重耐药细菌并减轻 AMR。这一成果源于对七种 MCHPN 的结构、抗菌活性和生物相容性的分析,其中 MCHPN7 的阳离子基团、阴离子基团和长烷基链的比例为 27:59:14,成为主要候选物质。重要的是,由于不同物种的膜电位存在固有差异,加上其纳米尺寸(6 - 15 nm)和高亲水性(Kow = 0.04),MCHPN7 在体外和体内对哺乳动物细胞表现出卓越的选择性杀菌效果(选择性指数 > 564)。通过诱导物理膜破坏,MCHPN7 能有效消灭耐抗生素细菌,并显著延缓细菌耐药性的产生。作为一种涂层,MCHPN7 使原本惰性的表面具有了阻碍生物膜形成和减轻小鼠模型感染相关免疫反应的能力。这项研究预示着生物相容性聚合物纳米粒子的出现,对我们正在进行的抗 AMR 斗争具有重要意义。意义声明:抗菌素耐药性(AMR)的不断升级已被公认为是对全球健康的最大威胁之一。因此,我们设计并合成了一系列具有选择性抗菌作用的混合电荷超支化聚合物纳米粒子(MCHPNs)。由于不同物种之间固有的膜电位差异和高亲水性(Kow = 0.04),最佳纳米粒子对哺乳动物细胞表现出卓越的选择性杀菌效果(选择性指数大于 564),并能显著延缓细菌耐药性的产生。重要的是,它们赋予了表面阻碍生物膜形成和减轻感染相关免疫反应的能力。此外,上述研究结果重点解决了大流行后的 AMR 问题,必将引起学术界和工业界研究人员的关注。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

Acta Biomaterialia
工程技术-材料科学:生物材料
CiteScore
16.80
自引率
3.10%
发文量
776
审稿时长
30 days
期刊介绍:
Acta Biomaterialia is a monthly peer-reviewed scientific journal published by Elsevier. The journal was established in January 2005. The editor-in-chief is W.R. Wagner (University of Pittsburgh). The journal covers research in biomaterials science, including the interrelationship of biomaterial structure and function from macroscale to nanoscale. Topical coverage includes biomedical and biocompatible materials.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


