Formation of cyclopenta[c]quinolines through visible-light-induced photoredox cascade bis-annulations of 1,7-enynes with sulfoxonium ylides†
IF 4.6
Q2 MATERIALS SCIENCE, BIOMATERIALS
引用次数: 0
Abstract
A novel visible-light-driven photoredox-catalyzed cascade bicyclization of 1,7-enynes with aqueous sulfoxonium ylides is reported. The reaction is highly chemoselective with three new C–C bonds, two new rings, and an all-carbon quaternary stereocenter constructed in a one-pot fashion. This mild protocol features a remarkably broad substrate scope with good functional group tolerance, providing a general and practical approach to access various cyclopenta[c]quinolines.
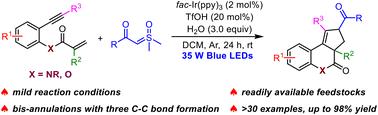
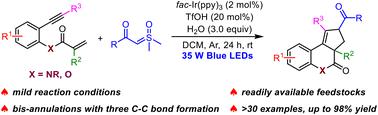
在可见光诱导下,1,7-烯炔与亚磺鎓酰化物发生光氧化级联反应,生成环戊并[c]喹啉。
本研究报道了一种新型可见光驱动的光氧催化 1,7-enynes 与水性锍酰化物的级联双环化反应。该反应具有高度化学选择性,能以一锅方式生成三个新的 C-C 键、两个新的环和一个全碳四元立体中心。这种温和的反应方案具有非常广泛的底物范围和良好的官能团耐受性,为获得各种环戊并[c]喹啉提供了一种通用而实用的方法。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文

 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


