Reversed Charge Transfer Enables Dual Active Sites on Ir/hBN for Synergistic N2O Valorization and Propane Selective Oxidation
IF 11.3
1区 化学
Q1 CHEMISTRY, PHYSICAL
引用次数: 0
Abstract
Valorization of nitrous oxide (N2O) as a mild oxygen source for light alkanes presents a promising and economical method for mitigating global warming. However, activating N2O and alkane together often leads to overoxidation and poor selectivity of the products. To disentangle the trade-off between activity and selectivity, herein, an Ir-based hexagonal boron nitride (hBN) catalyst was synthesized to obtain a reversed charge transfer (RCT) from the support to metal centers, forming dual active sites on Ir clusters and the separation of redox centers, as determined via operando near-ambient-pressure X-ray photoelectron spectroscopy (NAP-XPS) and density functional theory (DFT) calculations. Ir/hBN demonstrated a high N2O conversion (99.5%) and syngas yield (95.9 mol of CO kgcat–1 h–1 and 41.9 mol of H2 kgcat–1 h–1) during the selective oxidation of propane (C3H8) at 450 °C. The electron-rich Ir interfacial perimeter sites (Irδ−) enhance N2O adsorption and N–O bond dissociation to produce active O*; however, facial metallic Ir0 sites effectively facilitate C3H8 activation, including dehydrogenation and cracking. The separation of H* and O* intermediates, along with the frustrated H*/O* spillover, effectively facilitates the formation of H2. The *CH2 intermediate from C3H8 breakage migrates and reacts with O* bound to Ir interfacial sites, where it is oxidized to CO32– and subsequently liberates CO. This study provides mechanistic insights into the O element valorization from N2O with synergetic enhancement in selective oxidation of light alkanes.
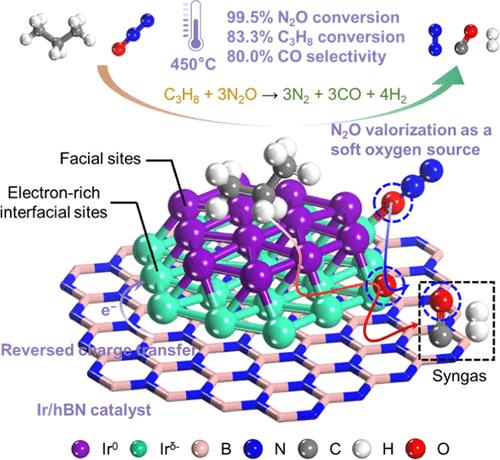
反向电荷转移使 Ir/hBN 上的双活性位协同实现 N2O 有效化和丙烷选择性氧化
将一氧化二氮(N2O)作为轻质烷烃的温和氧源进行有效利用,是减缓全球变暖的一种既有前景又经济的方法。然而,将一氧化二氮和烷烃一起活化往往会导致过氧化和产物选择性差。为了解决活性和选择性之间的权衡问题,本文合成了一种以 Ir 为基质的六方氮化硼(hBN)催化剂,通过操作性近常压 X 射线光电子能谱(NAP-XPS)和密度泛函理论(DFT)计算,获得了从载体到金属中心的反向电荷转移(RCT),在 Ir 簇上形成了双活性位点,并分离了氧化还原中心。在 450 °C 下选择性氧化丙烷(C3H8)时,Ir/hBN 表现出了很高的 N2O 转化率(99.5%)和合成气产量(95.9 摩尔 CO kgcat-1 h-1 和 41.9 摩尔 H2 kgcat-1 h-1)。富含电子的 Ir 介面周缘位点(Irδ-)增强了对 N2O 的吸附和 N-O 键的解离,从而产生活性 O*;然而,面金属 Ir0 位点则有效地促进了 C3H8 的活化,包括脱氢和裂解。H* 和 O* 中间体的分离以及受挫的 H*/O* 溢出有效地促进了 H2 的形成。C3H8 断裂产生的 *CH2 中间体迁移并与结合在 Ir 界面位点上的 O* 发生反应,被氧化成 CO32-,随后释放出 CO。这项研究从机理上揭示了如何从 N2O 中实现 O 元素的价值化,并在轻质烷烃的选择性氧化过程中实现协同增效。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

ACS Catalysis
CHEMISTRY, PHYSICAL-
CiteScore
20.80
自引率
6.20%
发文量
1253
审稿时长
1.5 months
期刊介绍:
ACS Catalysis is an esteemed journal that publishes original research in the fields of heterogeneous catalysis, molecular catalysis, and biocatalysis. It offers broad coverage across diverse areas such as life sciences, organometallics and synthesis, photochemistry and electrochemistry, drug discovery and synthesis, materials science, environmental protection, polymer discovery and synthesis, and energy and fuels.
The scope of the journal is to showcase innovative work in various aspects of catalysis. This includes new reactions and novel synthetic approaches utilizing known catalysts, the discovery or modification of new catalysts, elucidation of catalytic mechanisms through cutting-edge investigations, practical enhancements of existing processes, as well as conceptual advances in the field. Contributions to ACS Catalysis can encompass both experimental and theoretical research focused on catalytic molecules, macromolecules, and materials that exhibit catalytic turnover.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


