Hyaluronic acid-alginate hydrogel stimulates the differentiation of neonatal mouse testicular cells into hepatocyte-like and other cell lineages in three-dimensional culture
Abstract
Background information
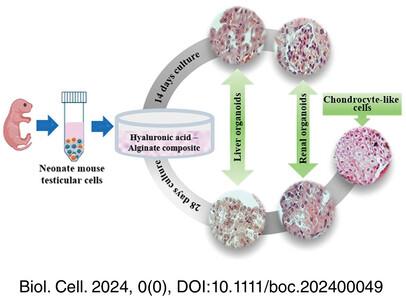
Extracellular matrix (ECM)-derived hydrogels are frequently used in three-dimensional (3D) cell culture and organoid formation in several tissues. However, in the 3D cultivation of testicular cells, the hyaluronic acid (HA) hydrogel has not received as much attention. This study examined the effects of three distinct composites, including HA-alginate (HA-Alg), HA-alginate-collagen (HA-Alg-Col), and HA-alginate-decellularized ECM (HA-Alg-dECM), on mouse testicular cell culture and in vitro spermatogenesis.
Methods
For the creation of composites, the concentration of biomaterials used was 0.5% HA, 1% alginate, 2.5 mg/mL collagen, and 25 mg/mL dECM derived from the testicles of Rams. After 3D culture of 5 days post-partum (dpp) mouse testicular cells for 14 days, HA-Alg was selected as a superior composite due to the greater number and size of the produced organoids. Then, cell culture was rerun by HA-Alg for 14 days, which was later extended for an additional 28 days. In addition, the 3D culture of 10 dpp mouse testicular cells was used to compare with 5 dpp mice on day 14. The morphology and gene expression were analyzed using appropriate techniques.
Results
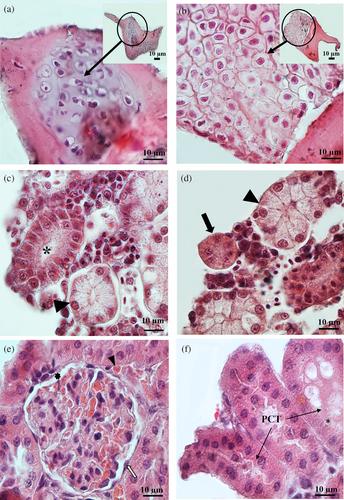
On day 14, the HA-Alg hydrogel showed significantly more organoids in terms of size and number than the other two groups (p < 0.05); nevertheless, none of the groups showed the expected signs of testis organoids. Remarkably, on day 14, the histology and immunostaining tests revealed features of hepatocyte-like cells (HLCs) and albumin production as a marker of HLC functionality. Furthermore, the analysis of gene expression verified the significant expression of angiogenesis markers (p < 0.01). After the extended culture to 28 days, 5 dpp testicular cells once more differentiated into erythrocytes and HLCs, while a small number of organoids showed the characteristic of renal cells. Cell culture of 10 dpp mice for 14 days showed a wide range of cell lineages, including renal, glandular, chondrocyte, and hepatocyte-like cells in comparison to the 5 dpp mice.
Conclusion and significance
While the HA-Alg composite did not support spermatogenesis in the 3D culture of mouse testicular cells, it demonstrated an unpredicted potential for promoting the differentiation of neonate mouse testicular cells into HLC, erythrocytes, and other cell lineages.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


