Bioengineering of syrbactin megasynthetases for immunoproteasome inhibitor production
IF 19.1
1区 化学
Q1 CHEMISTRY, MULTIDISCIPLINARY
引用次数: 0
Abstract
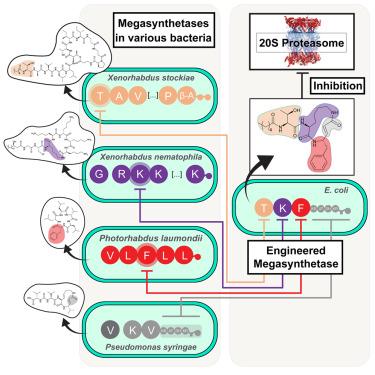
The natural product (NP) class of syrbactins are potent proteasome inhibitors produced by hybrids of non-ribosomal peptide synthetases (NRPSs) and polyketide synthases (PKSs). Here, we describe the stepwise reassembly of an entire NRPS/PKS hybrid to produce a new syrbactin derivative by utilizing the recently described “eXchange Unit between Thiolation domains” (XUTs) approach. Remarkably, XUT-based engineering allowed the direct assembly of PKS and NRPS modules to introduce an α,β-unsaturated Michael system in a macrolactam moiety, which represents the inhibitory warhead of syrbactins. The novel derivative was produced in E. coli, isolated, and examined for its ability to inhibit yeast (yCP), human constitutive (cCP), and immunoproteasome (iCP). The engineered NP maintained the inhibitory activities of the syrbactin class but, due to rational modifications, inhibited iCP most strongly. Moreover, analysis of the crystal structure of yCP in complex with the derivative revealed further design strategies for even more specific iCP inhibition.
用于生产免疫蛋白酶体抑制剂的西林杆菌巨合成酶生物工程
天然产物 (NP) 类西布曲明是由非核糖体肽合成酶 (NRPS) 和多肽合成酶 (PKS) 杂交产生的强效蛋白酶体抑制剂。在这里,我们描述了利用最近描述的 "硫化结构域之间的交换单元"(XUTs)方法,逐步重新组装整个 NRPS/PKS 杂交体,从而产生一种新的 syrbactin 衍生物。值得注意的是,基于 XUT 的工程学方法可以直接组装 PKS 和 NRPS 模块,从而在大内酰胺分子中引入一个 α、β-不饱和迈克尔系统,该系统代表了 syrbactins 的抑制性弹头。这种新型衍生物在大肠杆菌中产生、分离并检测了其抑制酵母(yCP)、人组成型(cCP)和免疫蛋白酶体(iCP)的能力。改造后的 NP 保持了 syrbactin 类的抑制活性,但由于进行了合理的修饰,对 iCP 的抑制作用最强。此外,通过分析 yCP 与衍生物复合物的晶体结构,还发现了进一步的设计策略,以实现更特异的 iCP 抑制作用。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

Chem
Environmental Science-Environmental Chemistry
CiteScore
32.40
自引率
1.30%
发文量
281
期刊介绍:
Chem, affiliated with Cell as its sister journal, serves as a platform for groundbreaking research and illustrates how fundamental inquiries in chemistry and its related fields can contribute to addressing future global challenges. It was established in 2016, and is currently edited by Robert Eagling.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


