Nickel-catalysed reductive C–N bond cross-coupling between aryl halides and N-chloroamides†
IF 2.9
3区 化学
Q1 CHEMISTRY, ORGANIC
引用次数: 0
Abstract
A method for the direct synthesis of N-aryl lactams and amides with aryl halides and N-chloroamides through a Ni-catalyzed reductive C–N coupling reaction has been developed. The reaction features the advantages of mild conditions, good functional group tolerance and broad substrate scope including drug-derived substrates, and also provided direct access to the key synthetic intermediates for some bioactive molecules, suggesting the practicability of this method. Finally, DFT calculations were performed to shed further light on the reaction mechanism and it was found that an amidyl radical might be involved.
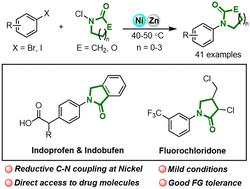
镍催化的芳基卤化物与 N-氯酰胺之间的还原性 C-N 键交叉耦合。
通过镍催化的还原性 C-N 偶联反应,开发了一种用芳基卤化物和 N-氯酰胺直接合成 N-芳基内酰胺和酰胺的方法。该反应具有条件温和、官能团耐受性好、底物范围广(包括药物衍生底物)等优点,还为一些生物活性分子提供了直接获得关键合成中间体的途径,表明了该方法的实用性。最后,通过 DFT 计算进一步揭示了反应机理,发现酰胺基可能参与了反应。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

Organic & Biomolecular Chemistry
化学-有机化学
CiteScore
5.50
自引率
9.40%
发文量
1056
审稿时长
1.3 months
期刊介绍:
The international home of synthetic, physical and biomolecular organic chemistry.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


