Mechanism of allosteric inhibition of human p97/VCP ATPase and its disease mutant by triazole inhibitors
IF 5.9
2区 化学
Q1 CHEMISTRY, MULTIDISCIPLINARY
引用次数: 0
Abstract
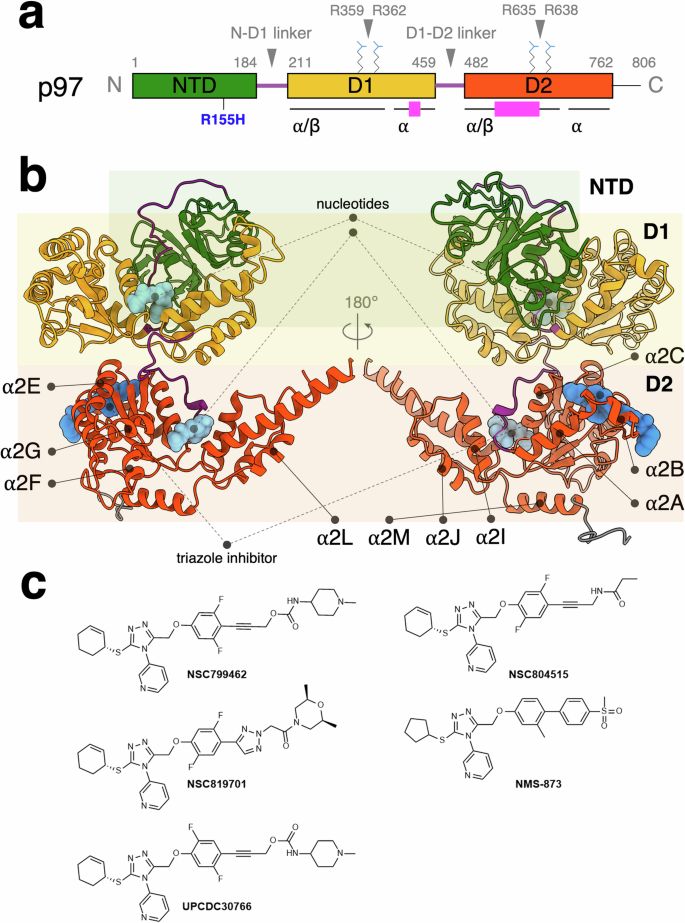
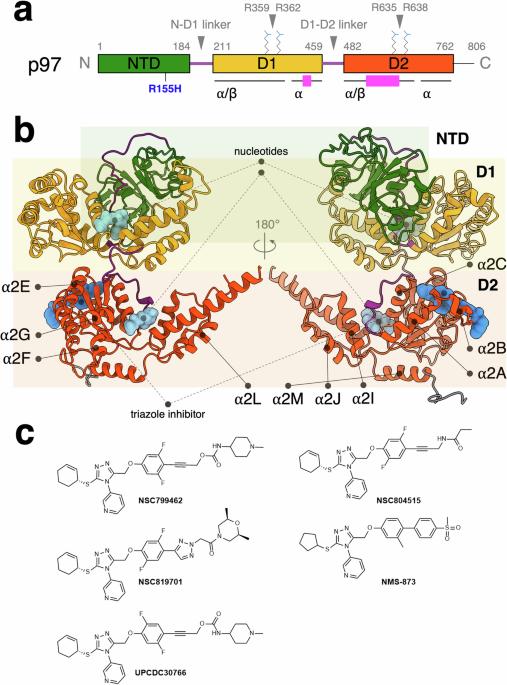
Human p97 ATPase is crucial in various cellular processes, making it a target for inhibitors to treat cancers, neurological, and infectious diseases. Triazole allosteric p97 inhibitors have been demonstrated to match the efficacy of CB-5083, an ATP-competitive inhibitor, in cellular models. However, the mechanism is not well understood. This study systematically investigates the structures of new triazole inhibitors bound to both wild-type and disease mutant forms of p97 and measures their effects on function. These inhibitors bind at the interface of the D1 and D2 domains of each p97 subunit, shifting surrounding helices and altering the loop structures near the C-terminal α2 G helix to modulate domain-domain communications. A key structural moiety of the inhibitor affects the rotameric conformations of interacting side chains, indirectly modulating the N-terminal domain conformation in p97 R155H mutant. The differential effects of inhibitor binding to wild-type and mutant p97 provide insights into drug design with enhanced specificity, particularly for oncology applications. Human p97 ATPase, a critical drug target for neurodegenerative disorders and cancers, can be allosterically inhibited by triazole-based inhibitors. In this study, the authors investigate the structure and functions of newly designed triazole inhibitors in both wild-type and disease mutant forms of p97 to elucidate the previously unexplored inhibitory mechanisms, shedding new light on the design concept for p97 allosteric inhibitors.
三唑类抑制剂对人类 p97/VCP ATPase 及其疾病突变体的异位抑制机制。
人类 p97 ATPase 在各种细胞过程中起着关键作用,因此成为治疗癌症、神经疾病和传染性疾病抑制剂的靶点。在细胞模型中,三唑异构 p97 抑制剂已被证明与 ATP 竞争性抑制剂 CB-5083 的疗效相当。然而,人们对其机制还不甚了解。本研究系统地研究了与野生型和疾病突变型 p97 结合的新型三唑抑制剂的结构,并测量了它们对功能的影响。这些抑制剂结合在每个 p97 亚基的 D1 和 D2 结构域的界面上,移动周围的螺旋并改变 C 端 α2 G 螺旋附近的环结构,从而调节结构域与结构域之间的通讯。抑制剂的一个关键结构分子会影响相互作用侧链的旋转构象,从而间接调节 p97 R155H 突变体的 N 端结构域构象。抑制剂与野生型和突变型 p97 结合的不同效应为设计具有更强特异性的药物(尤其是肿瘤应用药物)提供了启示。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊
Communications Chemistry
Chemistry-General Chemistry
CiteScore
7.70
自引率
1.70%
发文量
146
审稿时长
13 weeks
期刊介绍:
Communications Chemistry is an open access journal from Nature Research publishing high-quality research, reviews and commentary in all areas of the chemical sciences. Research papers published by the journal represent significant advances bringing new chemical insight to a specialized area of research. We also aim to provide a community forum for issues of importance to all chemists, regardless of sub-discipline.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


