Comparative safety of oral Janus kinase inhibitors versus dupilumab in patients with atopic dermatitis: A population-based cohort study
IF 11.4
1区 医学
Q1 ALLERGY
引用次数: 0
Abstract
Background
Systemic Janus kinase inhibitors (JAKi) and dupilumab both have emerged as promising therapeutics for atopic dermatitis (AD). Dupilumab has a favorable safety profile, but oral JAKi therapy has been established in other diseases that carry potential comorbid susceptibilities that influence safety.
Objective
We sought to provide real-world evidence of the comparative safety of oral JAKi versus dupilumab in patients with AD.
Methods
The study used observational data from multiple healthcare organizations in the US. Patients with AD treated with either oral JAKi (upadacitinib, abrocitinib, and baricitinib) or dupilumab were enrolled. The 2 treatment groups were propensity score matched 1:1 on the basis of demographics, comorbidities, and prior medications. Safety outcomes within 2 years after the initiation of medications were measured by hazard ratios (HRs) with 95% confidence intervals (CIs).
Results
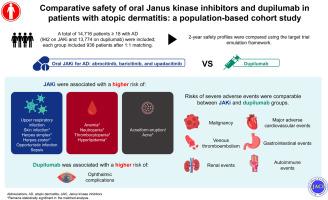
A total of 14,716 patients were included, with 942 patients treated with oral JAKi and 13,774 with dupilumab. The 2 treatment groups respectively included 938 patients after matching. Treatment with oral JAKi was not associated with increased risks of mortality, malignancies, major adverse cardiovascular events, venous thromboembolism, renal events, or serious gastrointestinal events. However, patients receiving oral JAKi showed significantly higher risks of skin and subcutaneous tissue infection (HR = 1.35, 95% CI = 1.07-1.69), herpes infection (herpes simplex, HR = 1.64, 95% CI = 1.03-2.61; herpes zoster, HR = 2.51, 95% CI = 1.14-5.52), acne (HR = 2.09, 95% CI = 1.54-2.84), cytopenia (anemia, HR = 1.83, 95% CI = 1.39-2.41; neutropenia, HR = 4.02, 95% CI = 1.91-8.47; thrombocytopenia, HR = 1.76, 95% CI = 1.08-2.89), and hyperlipidemia (HR = 1.45, 95% CI = 1.09-1.92); the risk of ophthalmic complications was higher in those receiving dupilumab (HR = 1.49, 95% CI = 1.03-2.17).
Conclusion
Oral JAKi did not exhibit concerning safety issues in treating patients with AD but increased the risk of infections and abnormalities in laboratory findings. Long-term follow-up data are required to validate these results.
特应性皮炎患者口服 Janus 激酶抑制剂和杜匹单抗的安全性比较:一项基于人群的队列研究。
背景:全身性 Janus 激酶抑制剂(JAKi)和杜比单抗都已成为治疗特应性皮炎(AD)的有前途的疗法。尽管杜比鲁单抗具有良好的安全性,但口服 JAKi 已被用于其他疾病,而这些疾病具有影响安全性的潜在并发症:为口服 JAKi 在 AD 患者中的安全性提供实际证据:研究使用了 TriNetX(马萨诸塞州剑桥市)的观察数据。接受口服 JAKi(upadacitinib、abrocitinib 和 baricitinib)或 dupilumab 治疗的 AD 患者被纳入研究。两个治疗组根据人口统计学、合并症和既往用药情况进行了倾向得分匹配,比例为1:1。用危险比和95%置信区间来衡量用药后两年内的安全性结果:共纳入14716名患者,其中942名患者接受了口服JAKi治疗,13774名患者接受了dupilumab治疗。两个治疗组在匹配后共纳入938名患者。口服JAKi治疗与死亡率、恶性肿瘤、主要不良心血管事件、静脉血栓栓塞、肾脏事件或严重胃肠道事件的风险增加无关。然而,接受口服JAKi治疗的患者发生皮肤和皮下组织感染、疱疹感染、痤疮、全血细胞减少症和高脂血症的风险明显较高,而接受dupilumab治疗的患者发生眼科并发症的风险较高:这项研究发现,口服JAKi在治疗AD患者时并没有表现出令人担忧的安全性问题,但会增加感染和实验室异常的风险。需要长期随访数据来验证这些发现。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊
CiteScore
25.90
自引率
7.70%
发文量
1302
审稿时长
38 days
期刊介绍:
The Journal of Allergy and Clinical Immunology is a prestigious publication that features groundbreaking research in the fields of Allergy, Asthma, and Immunology. This influential journal publishes high-impact research papers that explore various topics, including asthma, food allergy, allergic rhinitis, atopic dermatitis, primary immune deficiencies, occupational and environmental allergy, and other allergic and immunologic diseases. The articles not only report on clinical trials and mechanistic studies but also provide insights into novel therapies, underlying mechanisms, and important discoveries that contribute to our understanding of these diseases. By sharing this valuable information, the journal aims to enhance the diagnosis and management of patients in the future.

 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


