Inhibition of BCAT1-mediated cytosolic leucine metabolism regulates Th17 responses via the mTORC1-HIF1α pathway
IF 9.5
2区 医学
Q1 BIOCHEMISTRY & MOLECULAR BIOLOGY
引用次数: 0
Abstract
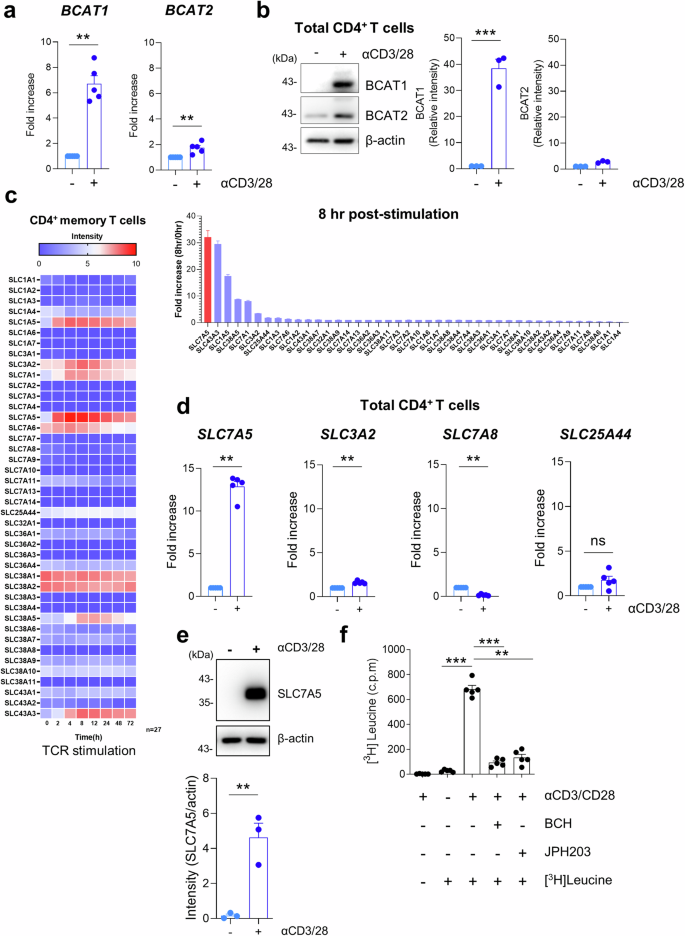
Branched-chain amino acids (BCAAs), particularly leucine, are indispensable AAs for immune regulation through metabolic rewiring. However, the molecular mechanism underlying this phenomenon remains unclear. Our investigation revealed that T-cell receptor (TCR)-activated human CD4+ T cells increase the expression of BCAT1, a cytosolic enzyme responsible for BCAA catabolism, and SLC7A5, a major BCAA transporter. This upregulation facilitates increased leucine influx and catabolism, which are particularly crucial for Th17 responses. Activated CD4+ T cells induce an alternative pathway of cytosolic leucine catabolism, generating a pivotal metabolite, β-hydroxy β-methylbutyric acid (HMB), by acting on BCAT1 and 4-hydroxyphenylpyruvate dioxygenase (HPD)/HPD-like protein (HPDL). Inhibition of BCAT1-mediated cytosolic leucine metabolism, either with BCAT1 inhibitor 2 (Bi2) or through BCAT1, HPD, or HPDL silencing using shRNA, attenuates IL-17 production, whereas HMB supplementation abrogates this effect. Mechanistically, HMB contributes to the regulation of the mTORC1-HIF1α pathway, a major signaling pathway for IL-17 production, by increasing the mRNA expression of HIF1α. This finding was corroborated by the observation that treatment with L-β-homoleucine (LβhL), a leucine analog and competitive inhibitor of BCAT1, decreased IL-17 production by TCR-activated CD4+ T cells. In an in vivo experimental autoimmune encephalomyelitis (EAE) model, blockade of BCAT1-mediated leucine catabolism, either through a BCAT1 inhibitor or LβhL treatment, mitigated EAE severity by decreasing HIF1α expression and IL-17 production in spinal cord mononuclear cells. Our findings elucidate the role of BCAT1-mediated cytoplasmic leucine catabolism in modulating IL-17 production via HMB-mediated regulation of mTORC1-HIF1α, providing insights into its relevance to inflammatory conditions. T-cell, a type of infection-fighting white blood cell, alter their metabolic process, relying heavily on amino acids, the building blocks of proteins. This study investigates how T cells use the amino acid leucine to power their response. Researchers conducted experiments with human T-cell and a mouse model of autoimmune disease, a condition where the body attacks its own cells. They studied how leucine’s metabolic process affects T-cell function. The study discovered that a specific process involving leucine’s metabolic pathway in T cells is vital for their ability to produce IL-17. Blocking a crucial enzyme reduced IL-17 production and eased symptoms in a mouse model of autoimmune disease. These findings underline the importance of leucine’s metabolic process in T-cell function and suggest a potential target for treating autoimmune diseases more effectively, offering hope for new treatments. This summary was initially drafted using artificial intelligence, then revised and fact-checked by the author.

抑制 BCAT1 介导的细胞膜亮氨酸代谢可通过 mTORC1-HIF1α 途径调节 Th17 反应。
支链氨基酸(BCAAs),尤其是亮氨酸,是通过新陈代谢重新布线进行免疫调节的不可或缺的氨基酸。然而,这一现象的分子机制仍不清楚。我们的研究发现,T 细胞受体(TCR)激活的人类 CD4+ T 细胞会增加 BCAT1(一种负责 BCAA 分解代谢的细胞膜酶)和 SLC7A5(一种主要的 BCAA 转运体)的表达。这种上调促进了亮氨酸流入和分解的增加,这对 Th17 反应尤为重要。活化的 CD4+ T 细胞通过作用于 BCAT1 和 4-hydroxyphenylpyruvate dioxygenase (HPD)/HPD-like protein (HPDL),诱导细胞膜亮氨酸分解代谢的替代途径,产生一种关键的代谢产物--β-羟基 β-甲基丁酸 (HMB)。使用 BCAT1 抑制剂 2(Bi2)或使用 shRNA 抑制 BCAT1、HPD 或 HPDL 可抑制 BCAT1 介导的细胞质亮氨酸代谢,从而减少 IL-17 的产生,而补充 HMB 则可消除这种影响。从机理上讲,HMB通过增加HIF1α的mRNA表达,有助于调节mTORC1-HIF1α通路(IL-17产生的主要信号通路)。用亮氨酸类似物和 BCAT1 竞争性抑制剂 L-β-高亮氨酸(LβhL)处理 TCR 激活的 CD4+ T 细胞,可减少 IL-17 的产生,这一观察结果证实了上述发现。在活体实验性自身免疫性脑脊髓炎(EAE)模型中,通过 BCAT1 抑制剂或 LβhL 处理阻断 BCAT1 介导的亮氨酸分解,可降低脊髓单核细胞中 HIF1α 的表达和 IL-17 的产生,从而减轻 EAE 的严重程度。我们的研究结果阐明了 BCAT1 介导的细胞质亮氨酸分解代谢在通过 HMB 介导的 mTORC1-HIF1α 调节 IL-17 生成过程中的作用,为其与炎症条件的相关性提供了见解。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

Experimental and Molecular Medicine
医学-生化与分子生物学
CiteScore
19.50
自引率
0.80%
发文量
166
审稿时长
3 months
期刊介绍:
Experimental & Molecular Medicine (EMM) stands as Korea's pioneering biochemistry journal, established in 1964 and rejuvenated in 1996 as an Open Access, fully peer-reviewed international journal. Dedicated to advancing translational research and showcasing recent breakthroughs in the biomedical realm, EMM invites submissions encompassing genetic, molecular, and cellular studies of human physiology and diseases. Emphasizing the correlation between experimental and translational research and enhanced clinical benefits, the journal actively encourages contributions employing specific molecular tools. Welcoming studies that bridge basic discoveries with clinical relevance, alongside articles demonstrating clear in vivo significance and novelty, Experimental & Molecular Medicine proudly serves as an open-access, online-only repository of cutting-edge medical research.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


